Staphylococcus aureus fur regulates the expression of virulence factors that contribute to the pathogenesis of pneumonia
- PMID: 20100857
- PMCID: PMC2849423
- DOI: 10.1128/IAI.01423-09
Staphylococcus aureus fur regulates the expression of virulence factors that contribute to the pathogenesis of pneumonia
Abstract
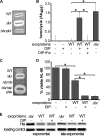
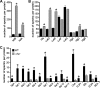
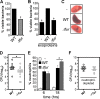
The tremendous success of Staphylococcus aureus as a pathogen is due to the controlled expression of a diverse array of virulence factors. The effects of host environments on the expression of virulence factors and the mechanisms by which S. aureus adapts to colonize distinct host tissues are largely unknown. Vertebrates have evolved to sequester nutrient iron from invading bacteria, and iron availability is a signal that alerts pathogenic microorganisms when they enter the hostile host environment. Consistent with this, we report here that S. aureus senses alterations in the iron status via the ferric uptake regulator (Fur) and alters the abundance of a large number of virulence factors. These Fur-mediated changes protect S. aureus against killing by neutrophils, and Fur is required for full staphylococcal virulence in a murine model of infection. A potential mechanistic explanation for the impact of Fur on virulence is provided by the observation that Fur coordinates the reciprocal expression of cytolysins and a subset of immunomodulatory proteins. More specifically, S. aureus lacking fur exhibits decreased expression of immunomodulatory proteins and increased expression of cytolysins. These findings reveal that Fur is involved in initiating a regulatory program that organizes the expression of virulence factors during the pathogenesis of S. aureus pneumonia.
Figures


Similar articles
-
Staphylococcus aureus Coordinates Leukocidin Expression and Pathogenesis by Sensing Metabolic Fluxes via RpiRc.mBio. 2016 Jun 21;7(3):e00818-16. doi: 10.1128/mBio.00818-16. mBio. 2016. PMID: 27329753 Free PMC article.
-
Differential expression and roles of Staphylococcus aureus virulence determinants during colonization and disease.mBio. 2015 Feb 17;6(1):e02272-14. doi: 10.1128/mBio.02272-14. mBio. 2015. PMID: 25691592 Free PMC article.
-
Fur is required for the activation of virulence gene expression through the induction of the sae regulatory system in Staphylococcus aureus.Int J Med Microbiol. 2011 Jan;301(1):44-52. doi: 10.1016/j.ijmm.2010.05.003. Epub 2010 Aug 12. Int J Med Microbiol. 2011. PMID: 20705504 Free PMC article.
-
Interplay between iron homeostasis and virulence: Fur and RyhB as major regulators of bacterial pathogenicity.Vet Microbiol. 2015 Aug 31;179(1-2):2-14. doi: 10.1016/j.vetmic.2015.03.024. Epub 2015 Apr 8. Vet Microbiol. 2015. PMID: 25888312 Review.
-
Immunopathogenesis of Staphylococcus aureus pulmonary infection.Semin Immunopathol. 2012 Mar;34(2):281-97. doi: 10.1007/s00281-011-0291-7. Epub 2011 Oct 31. Semin Immunopathol. 2012. PMID: 22037948 Free PMC article. Review.
Cited by
-
The heme-sensitive regulator SbnI has a bifunctional role in staphyloferrin B production by Staphylococcus aureus.J Biol Chem. 2019 Jul 26;294(30):11622-11636. doi: 10.1074/jbc.RA119.007757. Epub 2019 Jun 13. J Biol Chem. 2019. PMID: 31197035 Free PMC article.
-
Bacterial Responses to Iron Withholding by Calprotectin.Biochemistry. 2021 Nov 16;60(45):3337-3346. doi: 10.1021/acs.biochem.1c00572. Epub 2021 Nov 5. Biochemistry. 2021. PMID: 34739212 Free PMC article. Review.
-
Coordinated adaptation of Staphylococcus aureus to calprotectin-dependent metal sequestration.mBio. 2024 Jul 17;15(7):e0138924. doi: 10.1128/mbio.01389-24. Epub 2024 Jun 26. mBio. 2024. PMID: 38920392 Free PMC article.
-
Fpa (YlaN) is an iron(II) binding protein that functions to relieve Fur-mediated repression of gene expression in Staphylococcus aureus.mBio. 2024 Nov 13;15(11):e0231024. doi: 10.1128/mbio.02310-24. Epub 2024 Oct 23. mBio. 2024. PMID: 39440976 Free PMC article.
-
The transition from iron starvation to iron sufficiency as an important step in the progression of infection.Sci Prog. 2014;97(Pt 4):371-82. doi: 10.3184/003685014X14151846374739. Sci Prog. 2014. PMID: 25638949 Free PMC article. Review.
References
-
- Allard, M., H. Moisan, E. Brouillette, A. L. Gervais, M. Jacques, P. Lacasse, M. S. Diarra, and F. Malouin. 2006. Transcriptional modulation of some Staphylococcus aureus iron-regulated genes during growth in vitro and in a tissue cage model in vivo. Microbes Infect. 8:1679-1690. - PubMed
-
- Anderson, K. L., C. Roberts, T. Disz, V. Vonstein, K. Hwang, R. Overbeek, P. D. Olson, S. J. Projan, and P. M. Dunman. 2006. Characterization of the Staphylococcus aureus heat shock, cold shock, stringent, and SOS responses and their effects on log-phase mRNA turnover. J. Bacteriol. 188:6739-6756. - PMC - PubMed
-
- Beasley, F. C., E. D. Vines, J. C. Grigg, Q. Zheng, S. Liu, G. A. Lajoie, M. E. Murphy, and D. E. Heinrichs. 2009. Characterization of staphyloferrin A biosynthetic and transport mutants in Staphylococcus aureus. Mol. Microbiol. 72:947-963. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- AI69233/AI/NIAID NIH HHS/United States
- R01 AI069233/AI/NIAID NIH HHS/United States
- AI073780/AI/NIAID NIH HHS/United States
- F32 AI074278/AI/NIAID NIH HHS/United States
- U54 AI057157/AI/NIAID NIH HHS/United States
- T32 AI007474/AI/NIAID NIH HHS/United States
- T32 GM07347/GM/NIGMS NIH HHS/United States
- AI073843/AI/NIAID NIH HHS/United States
- F32 AI071487/AI/NIAID NIH HHS/United States
- R01 AI073843/AI/NIAID NIH HHS/United States
- AI071487/AI/NIAID NIH HHS/United States
- T32 AI-07474-13/AI/NIAID NIH HHS/United States
- T32 GM007347/GM/NIGMS NIH HHS/United States
- AI074278/AI/NIAID NIH HHS/United States
- R01 AI073780/AI/NIAID NIH HHS/United States
- T32 HL069765/HL/NHLBI NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources

