Polo-like kinase 4 kinase activity limits centrosome overduplication by autoregulating its own stability
- PMID: 20100909
- PMCID: PMC2813471
- DOI: 10.1083/jcb.200911102
Polo-like kinase 4 kinase activity limits centrosome overduplication by autoregulating its own stability
Abstract
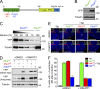
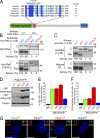
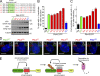
Accurate control of the number of centrosomes, the major microtubule-organizing centers of animal cells, is critical for the maintenance of genome integrity. Abnormalities in centrosome number can promote errors in spindle formation that lead to subsequent chromosome missegregation, and extra centrosomes are found in many cancers. Centrosomes are comprised of a pair of centrioles surrounded by amorphous pericentriolar material, and centrosome duplication is controlled by centriole replication. Polo-like kinase 4 (Plk4) plays a key role in initiating centriole duplication, and overexpression of Plk4 promotes centriole overduplication and the formation of extra centrosomes. Using chemical genetics, we show that kinase-active Plk4 is inherently unstable and targeted for degradation. Plk4 is shown to multiply self-phosphorylate within a 24-amino acid phosphodegron. Phosphorylation of multiple sites is required for Plk4 instability, indicating a requirement for a threshold level of Plk4 kinase activity to promote its own destruction. We propose that kinase-mediated, autoregulated instability of Plk4 self-limits Plk4 activity so as to prevent centrosome amplification.
Figures

References
-
- Burkard M.E., Randall C.L., Larochelle S., Zhang C., Shokat K.M., Fisher R.P., Jallepalli P.V. 2007. Chemical genetics reveals the requirement for Polo-like kinase 1 activity in positioning RhoA and triggering cytokinesis in human cells. Proc. Natl. Acad. Sci. USA. 104:4383–4388 10.1073/pnas.0701140104 - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

