Cancer chemoprevention locks onto a new polyamine metabolic target
- PMID: 20103728
- PMCID: PMC3574814
- DOI: 10.1158/1940-6207.CAPR-09-0252
Cancer chemoprevention locks onto a new polyamine metabolic target
Abstract
Ornithine decarboxylase has a relatively long history as a target for cancer chemoprevention and chemotherapy. Plym Forshell et al. report new evidence (beginning on p. 140 in this issue of the journal) indicating that spermidine synthase, a fellow enzyme of ornithine decarboxylase in polyamine metabolism, is transactivated in part by the MYC gene and is a potential target for chemoprevention of B-cell lymphomas.
Figures
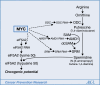
Comment on
-
Chemoprevention of B-cell lymphomas by inhibition of the Myc target spermidine synthase.Cancer Prev Res (Phila). 2010 Feb;3(2):140-7. doi: 10.1158/1940-6207.CAPR-09-0166. Epub 2010 Jan 26. Cancer Prev Res (Phila). 2010. PMID: 20103729
References
-
- Gerner EW, Meyskens FL., Jr. Polyamines and cancer: old molecules, new understanding. Nat Rev Cancer. 2004;4:781–92. - PubMed
-
- Plym Forshell TZ, Rimpi S, Nilsson JA. Chemoprevention of B-cell lymphomas by inhibition of the Myc target spermidine synthase. Cancer Prev Res. 2010;3:140–7. - PubMed
-
- Seiler N. Thirty years of polyamine-related approaches to cancer therapy. Retrospect and prospect. Part 1. Selective enzyme inhibitors. Curr Drug Targets. 2003;4:537–64. - PubMed
-
- Millward MJ, Joshua A, Kefford R, et al. Multi-centre Phase II trial of the polyamine synthesis inhibitor SAM486A (CGP48664) in patients with metastatic melanoma. Invest New Drugs. 2005;23:253–6. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

