Inhibition of lung fluid clearance and epithelial Na+ channels by chlorine, hypochlorous acid, and chloramines
- PMID: 20106988
- PMCID: PMC2843221
- DOI: 10.1074/jbc.M109.073981
Inhibition of lung fluid clearance and epithelial Na+ channels by chlorine, hypochlorous acid, and chloramines
Abstract
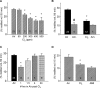
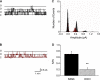
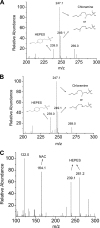
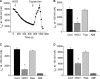
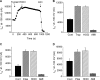
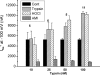
We investigated the mechanisms by which chlorine (Cl(2)) and its reactive byproducts inhibit Na(+)-dependent alveolar fluid clearance (AFC) in vivo and the activity of amiloride-sensitive epithelial Na(+) channels (ENaC) by measuring AFC in mice exposed to Cl(2) (0-500 ppm for 30 min) and Na(+) and amiloride-sensitive currents (I(Na) and I(amil), respectively) across Xenopus oocytes expressing human alpha-, beta-, and gamma-ENaC incubated with HOCl (1-2000 microm). Both Cl(2) and HOCl-derived products decreased AFC in mice and whole cell and single channel I(Na) in a dose-dependent manner; these effects were counteracted by serine proteases. Mass spectrometry analysis of the oocyte recording medium identified organic chloramines formed by the interaction of HOCl with HEPES (used as an extracellular buffer). In addition, chloramines formed by the interaction of HOCl with taurine or glycine decreased I(Na) in a similar fashion. Preincubation of oocytes with serine proteases prevented the decrease of I(Na) by HOCl, whereas perfusion of oocytes with a synthetic 51-mer peptide corresponding to the putative furin and plasmin cleaving segment in the gamma-ENaC subunit restored the ability of HOCl to inhibit I(Na). Finally, I(Na) of oocytes expressing wild type alpha- and gamma-ENaC and a mutant form of beta ENaC (S520K), known to result in ENaC channels locked in the open position, were not altered by HOCl. We concluded that HOCl and its reactive intermediates (such as organic chloramines) inhibit ENaC by affecting channel gating, which could be relieved by proteases cleavage.
Figures

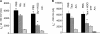
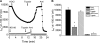
Similar articles
-
Alpha(1)-antitrypsin inhibits epithelial Na+ transport in vitro and in vivo.Am J Respir Cell Mol Biol. 2009 Sep;41(3):261-70. doi: 10.1165/rcmb.2008-0384OC. Epub 2009 Jan 8. Am J Respir Cell Mol Biol. 2009. PMID: 19131639 Free PMC article.
-
Regulation of alveolar epithelial Na+ channels by ERK1/2 in chlorine-breathing mice.Am J Respir Cell Mol Biol. 2012 Mar;46(3):342-54. doi: 10.1165/rcmb.2011-0309OC. Epub 2011 Oct 13. Am J Respir Cell Mol Biol. 2012. PMID: 21997487 Free PMC article.
-
Epithelial sodium channel inhibition by AMP-activated protein kinase in oocytes and polarized renal epithelial cells.J Biol Chem. 2005 May 6;280(18):17608-16. doi: 10.1074/jbc.M501770200. Epub 2005 Mar 7. J Biol Chem. 2005. PMID: 15753079
-
ENaC at the cutting edge: regulation of epithelial sodium channels by proteases.J Biol Chem. 2009 Jul 31;284(31):20447-51. doi: 10.1074/jbc.R800083200. Epub 2009 Apr 28. J Biol Chem. 2009. PMID: 19401469 Free PMC article. Review.
-
COVID-19 and Liquid Homeostasis in the Lung-A Perspective through the Epithelial Sodium Channel (ENaC) Lens.Cells. 2022 May 31;11(11):1801. doi: 10.3390/cells11111801. Cells. 2022. PMID: 35681496 Free PMC article. Review.
Cited by
-
Assessment of locomotion in chlorine exposed mice by computer vision and neural networks.J Appl Physiol (1985). 2012 Mar;112(6):1064-72. doi: 10.1152/japplphysiol.01023.2011. Epub 2011 Dec 29. J Appl Physiol (1985). 2012. PMID: 22207722 Free PMC article.
-
Integrative assessment of chlorine-induced acute lung injury in mice.Am J Respir Cell Mol Biol. 2012 Aug;47(2):234-44. doi: 10.1165/rcmb.2012-0026OC. Epub 2012 Mar 23. Am J Respir Cell Mol Biol. 2012. PMID: 22447970 Free PMC article.
-
Halogen-Induced Chemical Injury to the Mammalian Cardiopulmonary Systems.Physiology (Bethesda). 2021 Sep 1;36(5):272-291. doi: 10.1152/physiol.00004.2021. Physiology (Bethesda). 2021. PMID: 34431415 Free PMC article. Review.
-
Monocyte-derived alveolar macrophage apolipoprotein E participates in pulmonary fibrosis resolution.JCI Insight. 2020 Mar 12;5(5):e134539. doi: 10.1172/jci.insight.134539. JCI Insight. 2020. PMID: 32027623 Free PMC article.
-
Hyaluronan and halogen-induced airway hyperresponsiveness and lung injury.Ann N Y Acad Sci. 2020 Nov;1479(1):29-43. doi: 10.1111/nyas.14415. Epub 2020 Jun 23. Ann N Y Acad Sci. 2020. PMID: 32578230 Free PMC article. Review.
References
-
- Matalon S., O'Brodovich H. (1999) Annu. Rev. Physiol. 61, 627–661 - PubMed
-
- Song W., Matalon S. (2007) Am. J. Physiol. Lung Cell Mol. Physiol. 293, L855–LL858 - PubMed
-
- Matthay M. A., Folkesson H. G., Clerici C. (2002) Physiol. Rev. 82, 569–600 - PubMed
-
- Nielsen V. G., Duvall M. D., Baird M. S., Matalon S. (1998) Am. J. Physiol. 275, L1127–L1133 - PubMed
-
- Fang X., Song Y., Hirsch J., Galietta L. J., Pedemonte N., Zemans R. L., Dolganov G., Verkman A. S., Matthay M. A. (2006) Am. J. Physiol. Lung Cell Mol. Physiol. 290, L242–LL249 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

