Phenotypic definition of effector and memory T-lymphocyte subsets in mice chronically infected with Mycobacterium tuberculosis
- PMID: 20107011
- PMCID: PMC2849327
- DOI: 10.1128/CVI.00368-09
Phenotypic definition of effector and memory T-lymphocyte subsets in mice chronically infected with Mycobacterium tuberculosis
Abstract
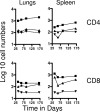
The bacterium Mycobacterium tuberculosis remains one of the world's most successful pathogens, a situation that is aggravated by the fact that the existing vaccine, Mycobacterium bovis BCG, is not effective in adults. As with any vaccine, the purpose of giving BCG vaccination is to establish a long-lived state of memory immunity, but whether this is successfully completely established is still unclear. It is generally accepted that memory T cells can be divided into central and effector memory populations by function and by phenotype; however, the majority of data supporting this division have been generated using transgenic mouse models or mice that have recovered from acute viral infections. Tuberculosis, on the other hand, represents a persistent, chronic state of immunity in which the presence of memory T cells is far less well defined. We show here that mice vaccinated with BCG or chronically infected with M. tuberculosis establish antigen-specific populations of cells within the lungs that predominantly express a cellular phenotype consistent with their being effector or effector memory cells. In contrast, cells with a central memory phenotype exist in much lower numbers in the lungs but can be found in significantly larger numbers in the spleen, where they may represent a potential reservoir. These data suggest that the effector-to-central-memory T-cell transition may well be minimal in these persisting mycobacterial infections, and they support a novel hypothesis that this may explain the fundamental basis of the failure of the BCG vaccine in humans.
Figures





Similar articles
-
Differential Mycobacterium bovis BCG vaccine-derived efficacy in C3Heb/FeJ and C3H/HeOuJ mice exposed to a clinical strain of Mycobacterium tuberculosis.Clin Vaccine Immunol. 2015 Jan;22(1):91-8. doi: 10.1128/CVI.00466-14. Epub 2014 Nov 12. Clin Vaccine Immunol. 2015. PMID: 25392011 Free PMC article.
-
Bacille Calmette-Guérin vaccination enhances human gamma delta T cell responsiveness to mycobacteria suggestive of a memory-like phenotype.J Immunol. 1998 Jul 15;161(2):1045-54. J Immunol. 1998. PMID: 9670986 Clinical Trial.
-
Heterologous Boost Following Mycobacterium bovis BCG Reduces the Late Persistent, Rather Than the Early Stage of Intranasal Tuberculosis Challenge Infection.Front Immunol. 2018 Oct 30;9:2439. doi: 10.3389/fimmu.2018.02439. eCollection 2018. Front Immunol. 2018. PMID: 30425711 Free PMC article.
-
Trying to See the Forest through the Trees: Deciphering the Nature of Memory Immunity to Mycobacterium tuberculosis.Front Immunol. 2018 Mar 8;9:461. doi: 10.3389/fimmu.2018.00461. eCollection 2018. Front Immunol. 2018. PMID: 29568298 Free PMC article. Review.
-
BCG - old workhorse, new skills.Curr Opin Immunol. 2017 Aug;47:8-16. doi: 10.1016/j.coi.2017.06.007. Epub 2017 Jul 15. Curr Opin Immunol. 2017. PMID: 28719821 Review.
Cited by
-
Mycobacterium bovis Wild-Type BCG or Recombinant BCG Secreting Murine IL-18 (rBCG/IL-18) Strains in Driving Immune Responses in Immunocompetent or Immunosuppressed Mice.Vaccines (Basel). 2022 Apr 14;10(4):615. doi: 10.3390/vaccines10040615. Vaccines (Basel). 2022. PMID: 35455364 Free PMC article.
-
Berberine governs NOTCH3/AKT signaling to enrich lung-resident memory T cells during tuberculosis.PLoS Pathog. 2023 Mar 7;19(3):e1011165. doi: 10.1371/journal.ppat.1011165. eCollection 2023 Mar. PLoS Pathog. 2023. PMID: 36881595 Free PMC article.
-
Tuberculosis vaccines and prevention of infection.Microbiol Mol Biol Rev. 2014 Dec;78(4):650-71. doi: 10.1128/MMBR.00021-14. Microbiol Mol Biol Rev. 2014. PMID: 25428938 Free PMC article. Review.
-
GI-19007, a Novel Saccharomyces cerevisiae-Based Therapeutic Vaccine against Tuberculosis.Clin Vaccine Immunol. 2017 Dec 5;24(12):e00245-17. doi: 10.1128/CVI.00245-17. Print 2017 Dec. Clin Vaccine Immunol. 2017. PMID: 29046306 Free PMC article.
-
Prime-boost approaches to tuberculosis vaccine development.Expert Rev Vaccines. 2012 Oct;11(10):1221-33. doi: 10.1586/erv.12.94. Expert Rev Vaccines. 2012. PMID: 23176655 Free PMC article. Review.
References
-
- Bevan, M. J. 2002. Immunology: remembrance of things past. Nature 420:748-749. - PubMed
-
- Bifani, P. J., B. Mathema, N. E. Kurepina, and B. N. Kreiswirth. 2002. Global dissemination of the Mycobacterium tuberculosis W-Beijing family strains. Trends Microbiol. 10:45-52. - PubMed
-
- Cavanagh, L. L., R. Bonasio, I. B. Mazo, C. Halin, G. Cheng, A. W. van der Velden, A. Cariappa, C. Chase, P. Russell, M. N. Starnbach, P. A. Koni, S. Pillai, W. Weninger, and U. H. von Andrian. 2005. Activation of bone marrow-resident memory T cells by circulating, antigen-bearing dendritic cells. Nat. Immunol. 6:1029-1037. - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials

