Early transcription from the maternal genome controlling blastomere integrity in mouse two-cell-stage embryos
- PMID: 20107036
- PMCID: PMC2867389
- DOI: 10.1152/ajpcell.00393.2009
Early transcription from the maternal genome controlling blastomere integrity in mouse two-cell-stage embryos
Abstract
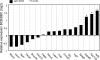
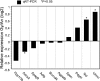
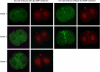
Blastomere cytofragmentation in mammalian embryos poses a significant problem in applied and clinical embryology. Mouse two-cell-stage embryos display strain-dependent differences in the rate of cytofragmentation, with a high rate observed in C3H/HeJ embryos and a lower rate observed in C57BL/6 embryos. The maternally inherited genome exerts the strongest effect on the process, with lesser effects mediated by the paternally inherited genome and the ooplasm. The effect of the maternal genome is transcription dependent and independent of the mitochondrial strain of origin. To identify molecular mechanisms that underlie cytofragmentation, we evaluated transcriptional activities of embryos possessing maternal pronuclei (mPN) of different origins. The mPN from C57BL/6 and C3H/HeJ strains directed specific transcription at the two-cell stage of mRNAs corresponding to 935 and 864 Affymetrix probe set IDs, respectively. Comparing transcriptomes of two-cell-stage embryos with different mPN revealed 64 transcribed genes with differential expression (1.4-fold or greater). Some of these genes occupy molecular pathways that may regulate cytofragmentation via a combination of effects related to apoptosis and effects on the cytoskeleton. These results implicate specific molecular mechanisms that may regulate cytofragmentation in early mammalian embryos. The most striking effect of mPN strain of origin on gene expression was on adenylate cyclase 2 (Adcy2). Treatment with dibutyryl cAMP (dbcAMP) elicits a high rate and severe form of cytofragmentation, and the effective dbcAMP concentration varies with maternal genotype. An activator of exchange proteins directly activated by cAMP (EPACs, or RAPGEF 3 and 4) 8-pCPT-2'-O-methyl-cAMP, elicits a high level of fragmentation while the PKA-specific activator N6-benzoyl-cAMP does not. Inhibition of A kinase anchor protein activities with st-Ht31 induces fragmentation. Inhibition of phosphatidylinositol 3-kinase signaling also induces fragmentation. These results reveal novel mechanisms by which maternal genotype affects cytofragmentation, including a system of opposing signaling pathways that most likely operate by controlling cytoskeletal function.
Figures


References
-
- Adenot PG, Mercier Y, Renard JP, Thompson EM. Differential H4 acetylation of paternal and maternal chromatin precedes DNA replication and differential transcriptional activity in pronuclei of 1-cell mouse embryos. Development 124: 4615–4625, 1997 - PubMed
-
- Alikani M, Calderon G, Tomkin G, Garrisi J, Kokot M, Cohen J. Cleavage anomalies in early human embryo and survival after prolonged culture in vitro. Hum Reprod 15: 2634–2643, 2000 - PubMed
-
- Antczak M, Van Blerkom J. Temporal and spatial aspects of fragmentation in early human embryos: possible effects on developmental competence and association with the differential elimination of regulatory proteins from polarized domains. Hum Reprod 14: 429–447, 1999 - PubMed
-
- Bedaiwy MA, Falcone T, Mohamed MS, Aleem AA, Sharma RK, Worley SE, Thornton J, Agarwal A. Differential growth of human embryos in vitro: role of reactive oxygen species. Fertil Steril 82: 593–600, 2004 - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

