Rapid, reversible modulation of blood-brain barrier P-glycoprotein transport activity by vascular endothelial growth factor
- PMID: 20107068
- PMCID: PMC2830816
- DOI: 10.1523/JNEUROSCI.5103-09.2010
Rapid, reversible modulation of blood-brain barrier P-glycoprotein transport activity by vascular endothelial growth factor
Abstract
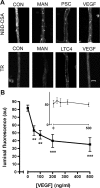
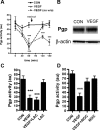
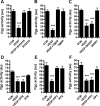
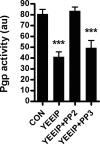
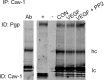
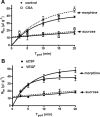
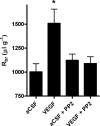
Increased brain expression of vascular endothelial growth factor (VEGF) is associated with neurological disease, brain injury, and blood-brain barrier (BBB) dysfunction. However, the specific effect of VEGF on the efflux transporter P-glycoprotein, a critical component of the BBB, is not known. Using isolated rat brain capillaries and in situ rat brain perfusion, we determined the effect of VEGF exposure on P-glycoprotein activity in vitro and in vivo. In isolated capillaries, VEGF acutely and reversibly decreased P-glycoprotein transport activity without decreasing transporter protein expression or opening tight junctions. This effect was blocked by inhibitors of the VEGF receptor flk-1 and Src kinase, but not by inhibitors of phosphatidylinositol-3-kinase or protein kinase C. VEGF also increased Tyr-14 phosphorylation of caveolin-1, and this was blocked by the Src inhibitor PP2. Pharmacological activation of Src kinase activity mimicked the effects of VEGF on P-glycoprotein activity and Tyr-14 phosphorylation of caveolin-1. In vivo, intracerebroventricular injection of VEGF increased brain distribution of P-glycoprotein substrates morphine and verapamil, but not the tight junction marker, sucrose; this effect was blocked by PP2. These findings indicate that VEGF decreases P-glycoprotein activity via activation of flk-1 and Src, and suggest Src-mediated phosphorylation of caveolin-1 may play a role in downregulation of P-glycoprotein activity. These findings also imply that P-glycoprotein activity is acutely diminished in pathological conditions associated with increased brain VEGF expression and that BBB VEGF/Src signaling could be targeted to acutely modulate P-glycoprotein activity and thus improve brain drug delivery.
Figures

Similar articles
-
Targeting blood-brain barrier sphingolipid signaling reduces basal P-glycoprotein activity and improves drug delivery to the brain.Proc Natl Acad Sci U S A. 2012 Sep 25;109(39):15930-5. doi: 10.1073/pnas.1203534109. Epub 2012 Sep 4. Proc Natl Acad Sci U S A. 2012. PMID: 22949658 Free PMC article.
-
Rapid loss of blood-brain barrier P-glycoprotein activity through transporter internalization demonstrated using a novel in situ proteolysis protection assay.J Cereb Blood Flow Metab. 2010 Sep;30(9):1593-7. doi: 10.1038/jcbfm.2010.117. Epub 2010 Jul 14. J Cereb Blood Flow Metab. 2010. PMID: 20628400 Free PMC article.
-
Deciphering mechanisms controlling placental artery endothelial cell migration stimulated by vascular endothelial growth factor.Endocrinology. 2010 Jul;151(7):3432-44. doi: 10.1210/en.2009-1305. Epub 2010 May 12. Endocrinology. 2010. PMID: 20463056 Free PMC article.
-
Functional expression and localization of P-glycoprotein at the blood brain barrier.Microsc Res Tech. 2002 Jun 1;57(5):365-80. doi: 10.1002/jemt.10090. Microsc Res Tech. 2002. PMID: 12112443 Review.
-
Modulation of P-glycoprotein at the blood-brain barrier: opportunities to improve central nervous system pharmacotherapy.Pharmacol Rev. 2008 Jun;60(2):196-209. doi: 10.1124/pr.107.07109. Epub 2008 Jun 17. Pharmacol Rev. 2008. PMID: 18560012 Free PMC article. Review.
Cited by
-
Drug transport across the blood-brain barrier.J Cereb Blood Flow Metab. 2012 Nov;32(11):1959-72. doi: 10.1038/jcbfm.2012.126. Epub 2012 Aug 29. J Cereb Blood Flow Metab. 2012. PMID: 22929442 Free PMC article. Review.
-
Neurovascular unit dysfunction with blood-brain barrier hyperpermeability contributes to major depressive disorder: a review of clinical and experimental evidence.J Neuroinflammation. 2013 Dec 1;10:142. doi: 10.1186/1742-2094-10-142. J Neuroinflammation. 2013. PMID: 24289502 Free PMC article. Review.
-
Gintonin facilitates brain delivery of donepezil, a therapeutic drug for Alzheimer disease, through lysophosphatidic acid 1/3 and vascular endothelial growth factor receptors.J Ginseng Res. 2021 Mar;45(2):264-272. doi: 10.1016/j.jgr.2019.12.002. Epub 2019 Dec 16. J Ginseng Res. 2021. PMID: 33841007 Free PMC article.
-
Targeting of multidrug-resistant human ovarian carcinoma cells with anti-P-glycoprotein antibody conjugates.Macromol Biosci. 2012 Apr;12(4):502-14. doi: 10.1002/mabi.201100350. Epub 2012 Jan 25. Macromol Biosci. 2012. PMID: 22278817 Free PMC article.
-
Pathways for small molecule delivery to the central nervous system across the blood-brain barrier.Perspect Medicin Chem. 2014 Jun 16;6:11-24. doi: 10.4137/PMC.S13384. eCollection 2014. Perspect Medicin Chem. 2014. PMID: 24963272 Free PMC article.
References
-
- Argyriou AA, Koltzenburg M, Polychronopoulos P, Papapetropoulos S, Kalofonos HP. Peripheral nerve damage associated with administration of taxanes in patients with cancer. Crit Rev Oncol Hematol. 2008;66:218–228. - PubMed
-
- Barakat S, Demeule M, Pilorget A, Regina A, Gingras D, Baggetto LG, Beliveau R. Modulation of p-glycoprotein function by caveolin-1 phosphorylation. J Neurochem. 2007;101:1–8. - PubMed
-
- Bauer B, Hartz AM, Miller DS. Tumor necrosis factor alpha and endothelin-1 increase P-glycoprotein expression and transport activity at the blood-brain barrier. Mol Pharmacol. 2007;71:667–675. - PubMed
-
- Bauer B, Hartz AM, Lucking JR, Yang X, Pollack GM, Miller DS. Coordinated nuclear receptor regulation of the efflux transporter, Mrp2, and the phase-II metabolizing enzyme, GSTpi, at the blood-brain barrier. J Cereb Blood Flow Metab. 2008;28:1222–1234. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous
