Duct system of the rabbit lacrimal gland: structural characteristics and role in lacrimal secretion
- PMID: 20107177
- PMCID: PMC2891459
- DOI: 10.1167/iovs.09-4687
Duct system of the rabbit lacrimal gland: structural characteristics and role in lacrimal secretion
Abstract
Purpose: To develop a nomenclature for the lacrimal duct system in the rabbit, based on the anatomic and structural characteristics of each duct segment, and to provide RT-PCR and immunofluorescence data to support the notion that the duct system plays important roles in lacrimal function.
Methods: Paraffin-embedded lacrimal glands (LGs) were stained with hematoxylin and eosin (H&E) and evaluated with a stereomicroscope. Cryosections of LG were stained with cresyl violet, and acinar cells and ductal epithelial cells were isolated from each duct segment by laser capture microdissection (LCM). mRNA levels from these cells were analyzed by real-time RT-PCR. Standard protocol was followed for immunofluorescence detection of ionic transporters.
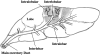
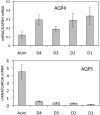
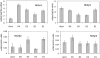
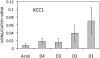
Results: The lacrimal duct system was divided into six segments on the basis of morphologic characteristics: the intercalated, intralobular, interlobular, intralobar, interlobar, and main excretory ducts. Although the morphologic features change incrementally along the entire duct system, the gene expression of ionic transporters and aquaporins, including AE3, AQP4, AQP5, CFTR, ClC2gamma, KCC1, NHE1, NKAalpha1, NKAbeta1, NKAbeta2, NKAbeta3, and NKCC1 varied greatly among duct segments. Immunofluorescence results were generally in accordance with the abundance of mRNAs along the acinus-duct axis.
Conclusions: Most LG research has focused on the acinar cells, with relatively little attention being paid to the lacrimal ducts. The lack of knowledge regarding the lacrimal ducts was so profound that a precise nomenclature had not been established for the duct system. The present data establish a nomenclature for each segment of the lacrimal duct system and provide evidence that ducts play critical roles in lacrimal secretion.
Figures


References
-
- Hisada M, Botelho SY. Membrane potentials of in situ lacrimal gland in the cat. Am J Physiol 1968;214:1262–1267 - PubMed
-
- Raina S, Preston GM, Guggino WB, Agre P. Molecular cloning and characterization of an aquaporin cDNA from salivary, lacrimal, and respiratory tissues. J Biol Chem 1995;270:1908–1912 - PubMed
-
- Hand A, Coleman R, Mazariegos MR, Lustmann J, Lotti LV. Endocytosis of proteins by salivary gland duct cells. J Dent Res 1987;66:412–419 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

