Cordycepin Suppresses Expression of Diabetes Regulating Genes by Inhibition of Lipopolysaccharide-induced Inflammation in Macrophages
- PMID: 20107539
- PMCID: PMC2803303
- DOI: 10.4110/in.2009.9.3.98
Cordycepin Suppresses Expression of Diabetes Regulating Genes by Inhibition of Lipopolysaccharide-induced Inflammation in Macrophages
Abstract
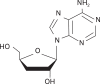
Background: It has been recently noticed that type 2 diabetes (T2D), one of the most common metabolic diseases, causes a chronic low-grade inflammation and activation of the innate immune system that are closely involved in the pathogenesis of T2D. Cordyceps militaris, a traditional medicinal mushroom, produces a component compound, cordycepin (3'-deoxyadenosine). Cordycepin has been known to have many pharmacological activities including immunological stimulating, anti-cancer, and anti-infection activities. The molecular mechanisms of cordycepin in T2D are not clear. In the present study, we tested the role of cordycepin on the anti-diabetic effect and anti-inflammatory cascades in LPS-stimulated RAW 264.7 cells.
Methods: We confirmed the levels of diabetes regulating genes mRNA and protein of cytokines through RT-PCR and western blot analysis and followed by FACS analysis for the surface molecules.
Results: Cordycepin inhibited the production of NO and pro-inflammatory cytokines such as IL-1beta, IL-6, and TNF-alpha in LPS-activated macrophages via suppressing protein expression of pro-inflammatory mediators. T2D regulating genes such as 11beta-HSD1 and PPARgamma were decreased as well as expression of co-stimulatory molecules such as ICAM-1 and B7-1/-2 were also decreased with the increment of its concentration. In accordance with suppressed pro-inflammatory cytokine production lead to inhibition of diabetic regulating genes in activated macrophages. Cordycepin suppressed NF-kappaB activation in LPS-activated macrophages.
Conclusion: Based on these observations, cordycepin suppressed T2D regulating genes through the inactivation of NF-kappaB dependent inflammatory responses and suggesting that cordycepin will provide potential use as an immunomodulatory agent for treating immunological diseases.
Keywords: cordycepin; immunomodulator; pro-inflammatory cytokines; type 2 diabetes.
Conflict of interest statement
The authors have no financial conflict of interest.
Figures




Similar articles
-
Cordycepin inhibits lipopolysaccharide-induced inflammation by the suppression of NF-kappaB through Akt and p38 inhibition in RAW 264.7 macrophage cells.Eur J Pharmacol. 2006 Sep 18;545(2-3):192-9. doi: 10.1016/j.ejphar.2006.06.047. Epub 2006 Jun 28. Eur J Pharmacol. 2006. PMID: 16899239
-
Anti-inflammatory effects of cordycepin via suppression of inflammatory mediators in BV2 microglial cells.Int Immunopharmacol. 2010 Dec;10(12):1580-6. doi: 10.1016/j.intimp.2010.09.011. Epub 2010 Oct 14. Int Immunopharmacol. 2010. PMID: 20937401
-
Anti-inflammatory effects of cordycepin in lipopolysaccharide-stimulated RAW 264.7 macrophages through Toll-like receptor 4-mediated suppression of mitogen-activated protein kinases and NF-κB signaling pathways.Drug Des Devel Ther. 2014 Oct 16;8:1941-53. doi: 10.2147/DDDT.S71957. eCollection 2014. Drug Des Devel Ther. 2014. PMID: 25342887 Free PMC article.
-
Docosahexaenoic acid attenuates LPS-stimulated inflammatory response by regulating the PPARγ/NF-κB pathways in primary bovine mammary epithelial cells.Res Vet Sci. 2017 Jun;112:7-12. doi: 10.1016/j.rvsc.2016.12.011. Epub 2017 Jan 5. Res Vet Sci. 2017. PMID: 28095338 Review.
-
Anti-inflammatory effects of cordycepin: A review.Phytother Res. 2020 Oct 8. doi: 10.1002/ptr.6890. Online ahead of print. Phytother Res. 2020. PMID: 33090621 Review.
Cited by
-
Dietary Phytochemicals: Natural Swords Combating Inflammation and Oxidation-Mediated Degenerative Diseases.Oxid Med Cell Longev. 2016;2016:5137431. doi: 10.1155/2016/5137431. Epub 2016 Sep 19. Oxid Med Cell Longev. 2016. PMID: 27721914 Free PMC article. Review.
-
Cordycepin prevented IL-β-induced expression of inflammatory mediators in human osteoarthritis chondrocytes.Int Orthop. 2014 Jul;38(7):1519-26. doi: 10.1007/s00264-013-2219-4. Epub 2013 Dec 12. Int Orthop. 2014. PMID: 24346509 Free PMC article.
-
Trends in the Immunomodulatory Effects of Cordyceps militaris: Total Extracts, Polysaccharides and Cordycepin.Front Pharmacol. 2020 Nov 30;11:575704. doi: 10.3389/fphar.2020.575704. eCollection 2020. Front Pharmacol. 2020. PMID: 33328984 Free PMC article. Review.
-
Cordycepin for Health and Wellbeing: A Potent Bioactive Metabolite of an Entomopathogenic Cordyceps Medicinal Fungus and Its Nutraceutical and Therapeutic Potential.Molecules. 2020 Jun 12;25(12):2735. doi: 10.3390/molecules25122735. Molecules. 2020. PMID: 32545666 Free PMC article. Review.
-
Structural and pharmacological insights into cordycepin for neoplasms and metabolic disorders.Front Pharmacol. 2024 Jun 17;15:1367820. doi: 10.3389/fphar.2024.1367820. eCollection 2024. Front Pharmacol. 2024. PMID: 38953102 Free PMC article. Review.
References
-
- Ogden C, Carroll M, Curtin L, McDowell M, Tabak C, Flegal K. Prevalence of overweight and obesity in the United States, 1999-2004. JAMA. 2006;295:1549–1555. - PubMed
-
- Li W, Zheng H, Bukuru J, De Kimpe N. Natural medicines used in the traditional Chinese medical system for therapy of diabetes mellitus. J Ethnopharmacol. 2004;92:1–21. - PubMed
-
- DeFronzo R. Pathogenesis of NIDDM. A balanced overview. Diabetes care. 1992;15:318–368. - PubMed
-
- sjöholm A, Nyström T. Endothelial inflammation in insulin resistance. Lancet. 2005;365:610–612. - PubMed
LinkOut - more resources
Full Text Sources
Miscellaneous

