Antidepressant interactions with the NMDA NR1-1b subunit
- PMID: 20107576
- PMCID: PMC2809336
- DOI: 10.1155/2008/474205
Antidepressant interactions with the NMDA NR1-1b subunit
Abstract
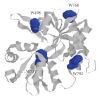
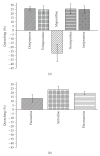
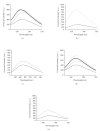
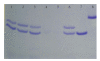
The targets for tricyclic antidepressants (TCAs), selective serotonin reuptake inhibitors (SSRIs), and selective norepinephrine reuptake inhibitors (SNRIs) are known to be the serotonin and norepinephrine transport (reuptake) proteins which are embedded in presynaptic nerve terminals and function to bring these neurotransmitters from the synaptic cleft back into the presynaptic neuron. Using a combination of intrinsic and extrinsic fluorescence quenching, Stern-Volmer analysis, and protease protection assays, we have shown that therapeutics from each of these three classes of antidepressants bind to the extracellular S1S2 domain of the NR1-1b subunit of the NMDA receptor. These results are in agreement with recent work from our lab demonstrating the interaction of antidepressants with the S1S2 domain of the GluR2 subunit of the AMPA receptor, another member of the ionotropic glutamate receptor subfamily, as well as work from other labs, and continue the discussion of the involvement of ionotropic glutamate receptors in depression.
Figures

Similar articles
-
Tricyclic antidepressants, but not the selective serotonin reuptake inhibitor fluoxetine, bind to the S1S2 domain of AMPA receptors.Arch Biochem Biophys. 2007 Feb 15;458(2):213-9. doi: 10.1016/j.abb.2006.12.004. Epub 2006 Dec 12. Arch Biochem Biophys. 2007. PMID: 17214956
-
Differential regulation of ionotropic glutamate receptors.Biophys J. 2007 Feb 15;92(4):1343-9. doi: 10.1529/biophysj.106.089896. Epub 2006 Nov 17. Biophys J. 2007. PMID: 17114218 Free PMC article.
-
Linking tricyclic antidepressants to ionotropic glutamate receptors.Biochem Biophys Res Commun. 2005 Jul 29;333(2):622-7. doi: 10.1016/j.bbrc.2005.05.114. Biochem Biophys Res Commun. 2005. PMID: 15946644
-
The role of selective serotonin reuptake inhibitors in preventing relapse of major depressive disorder.Ther Adv Psychopharmacol. 2018 Jan;8(1):49-58. doi: 10.1177/2045125317737264. Epub 2017 Nov 1. Ther Adv Psychopharmacol. 2018. PMID: 29344343 Free PMC article. Review.
-
Monoamine neurocircuitry in depression and strategies for new treatments.Prog Neuropsychopharmacol Biol Psychiatry. 2013 Aug 1;45:54-63. doi: 10.1016/j.pnpbp.2013.04.009. Epub 2013 Apr 19. Prog Neuropsychopharmacol Biol Psychiatry. 2013. PMID: 23602950 Review.
Cited by
-
Psychotropic Drugs for the Management of Chronic Pain and Itch.Pharmaceuticals (Basel). 2019 Jun 24;12(2):99. doi: 10.3390/ph12020099. Pharmaceuticals (Basel). 2019. PMID: 31238561 Free PMC article. Review.
-
Sertraline modulates hippocampal plasticity via sigma 1 receptors, cellular stress and neurosteroids.Transl Psychiatry. 2024 Nov 21;14(1):474. doi: 10.1038/s41398-024-03185-3. Transl Psychiatry. 2024. PMID: 39572523 Free PMC article.
-
Antidepressant drugs transactivate TrkB neurotrophin receptors in the adult rodent brain independently of BDNF and monoamine transporter blockade.PLoS One. 2011;6(6):e20567. doi: 10.1371/journal.pone.0020567. Epub 2011 Jun 7. PLoS One. 2011. PMID: 21666748 Free PMC article.
References
-
- Armstrong N, Gouaux E. Mechanisms for activation and antagonism of an AMPA-sensitive glutamate receptor: crystal structures of the GluR2 ligand binding core. Neuron. 2000;28(1):165–181. - PubMed
-
- Stoll L, Seguin S, Gentile L. Tricyclic antidepressants, but not the selective serotonin reuptake inhibitor fluoxetine, bind to the S1S2 domain of AMPA receptors. Archives of Biochemistry and Biophysics. 2007;458(2):213–219. - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

