Nesprins, but not sun proteins, switch isoforms at the nuclear envelope during muscle development
- PMID: 20108321
- PMCID: PMC3334500
- DOI: 10.1002/dvdy.22229
Nesprins, but not sun proteins, switch isoforms at the nuclear envelope during muscle development
Abstract
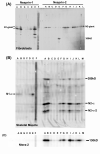
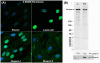
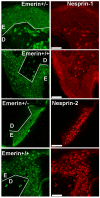
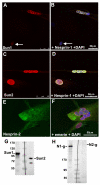
Nesprins are a family of nuclear transmembrane proteins anchored via Sun proteins to the nuclear membrane. Analysis of nesprins during human muscle development revealed an increase in nesprin-1-giant during early myogenesis in vitro. During the transition from immature to mature muscle fibres in vivo, nesprin-2 partly replaced nesprin-1 at the nuclear envelope and short nesprin isoforms became dominant. Sun1 and Sun2 proteins remained unchanged during this fibre maturation. In emerin-negative skin fibroblasts, nesprin-2-giant was relocated from the nuclear envelope to the cytoplasm, not to the endoplasmic reticulum, while nesprin-1 remained at the nuclear envelope. In emerin-negative keratinocytes lacking nesprin-1, nesprin-2 remained at the nuclear envelope. HeLa cell nuclear envelopes lacked nesprin-1, which was the dominant form in myoblasts, while a novel 130-kD nesprin-2 isoform dominated Ntera-2 cells. The results suggest the possibility of isoform-specific and tissue-specific roles for nesprins in nuclear positioning.
Copyright (c) 2010 Wiley-Liss, Inc.
Figures




References
-
- Apel ED, Lewis RM, Grady RM, Sanes JR. Syne-1, a dystrophin- and Klarsicht-related protein associated with synaptic nuclei at the neuromuscular junction. J. Biol. Chem. 2000;275:31986–31995. - PubMed
-
- Barbet JP, Thornell LE, Butler-Browne GS. Immunocytochemical characterisation of two generations of fibers during the development of the human quadriceps muscle. Mech Dev. 1991;35:3–11. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

