Trigeminal injury causes kappa opioid-dependent allodynic, glial and immune cell responses in mice
- PMID: 20109235
- PMCID: PMC2826348
- DOI: 10.1186/1744-8069-6-8
Trigeminal injury causes kappa opioid-dependent allodynic, glial and immune cell responses in mice
Abstract
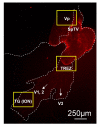
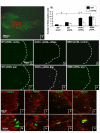
Background: The dynorphin-kappa opioid receptor (KOR) system regulates glial proliferation after sciatic nerve injury. Here, we investigated its role in cell proliferation following partial ligation of infraorbital nerve (pIONL), a model for trigeminal neuropathic pain. Mechanical allodynia was enhanced in KOR gene deleted mice (KOR-/-) compared to wild type mice. Using bromodeoxyuridine (BrdU) as a mitotic marker, we assessed cell proliferation in three different areas of the trigeminal afferent pathway: trigeminal nucleus principalis (Vp), trigeminal root entry zone (TREZ), and trigeminal ganglion (TG).
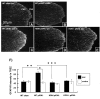
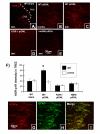
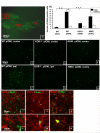
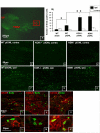
Results: In KOR-/- mice or norBNI-treated mice, the number of proliferating cells in the Vp was significantly less than in WT mice, whereas cell proliferation was enhanced in TREZ and TG. The majority of the proliferating cells were nestin positive stem cells or CD11b positive microglia in the Vp and macrophages in the TG. GFAP-positive astrocytes made a clear borderline between the CNS and the PNS in TREZ, and phosphorylated KOR staining (KOR-p) was detectable only in the astrocytes in CNS in WT mice but not in KOR-/- or norBNI-treated mice.
Conclusions: These results show that kappa opioid receptor system has different effects after pIONL in CNS and PNS: KOR activation promotes CNS astrocytosis and microglial or stem cell proliferation but inhibits macrophage proliferation in PNS. The trigeminal central root has a key role in the etiology and treatment of trigeminal neuralgia, and these newly identified responses may provide new targets for developing pain therapies.
Figures


References
-
- Xu M, Petraschka M, McLaughlin JP, Westenbroek RE, Caron MG, Lefkowitz RJ, Czyzyk TA, Pintar JE, Terman GW, Chavkin C. Neuropathic pain activates the endogenous kappa opioid system in mouse spinal cord and induces opioid receptor tolerance. J Neurosci. 2004;24:4576–4584. doi: 10.1523/JNEUROSCI.5552-03.2004. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

