Downregulation of FAK-related non-kinase mediates the migratory phenotype of human fibrotic lung fibroblasts
- PMID: 20109444
- PMCID: PMC2871958
- DOI: 10.1016/j.yexcr.2010.01.021
Downregulation of FAK-related non-kinase mediates the migratory phenotype of human fibrotic lung fibroblasts
Abstract
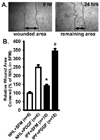
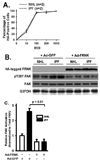
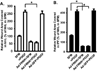
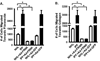
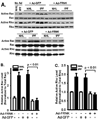
Fibroblast migration plays an important role in the normal wound healing process; however, dysregulated cell migration may contribute to the progressive formation of fibrotic lesions in the diseased condition. To examine the role of focal-adhesion-kinase (FAK)-related non-kinase (FRNK) in regulation of fibrotic lung fibroblast migration, we examined cell migration, FRNK expression, and activation of focal adhesion kinase (FAK) and Rho GTPase (Rho and Rac) in primary lung fibroblasts derived from both idiopathic pulmonary fibrosis (IPF) patients and normal human controls. Fibrotic (IPF) lung fibroblasts have increased cell migration when compared to control human lung fibroblasts. FRNK expression is significantly reduced in IPF lung fibroblasts, while activation of FAK, Rho and Rac is increased in IPF lung fibroblasts. Endogenous FRNK expression is inversely correlated with FAK activation and cell migration rate in IPF lung fibroblasts. Forced exogenous FRNK expression abrogates the increased cell migration, and blocked the activation of FAK and Rho GTPase (Rho and Rac), in IPF lung fibroblasts. These data for the first time provide evidence that downregulation of endogenous FRNK plays a role in promoting cell migration through FAK and Rho GTPase in fibrotic IPF lung fibroblasts.
Copyright 2010 Elsevier Inc. All rights reserved.
Figures

References
-
- King TE, Jr, Schwarz MI, Brown K, Tooze JA, Colby TV, Waldron JA, Jr, Flint A, Thurlbeck W, Chemiack RM. Idiopathic pulmonary fibrosis: relationship between histopathologic features and mortality. American Journal of Respiratory and Critical Care Medicine. 2001;164:1025–1032. - PubMed
-
- White ES, Thannickal VJ, Carskadon SL, Markwart S, Toews GB, Arenbert DA. Integrin alpha4beta1 regulates migration across basement membranes by lung fibroblasts: a role for phosphatase and tensin homologue deleted on chromosome 10. American Journal of Respiratory and Critical Care Medicine. 2003;168:436–442. - PMC - PubMed
-
- Parsons JT. Focal adhesion kinase: the first ten years. Journal of Cell Science. 2003;116:1409–1416. - PubMed
-
- Chen HC, Appeddu PA, Isoda H, Guan JL. Phosphorylation of tyrosine 397 in focal adhesion kinase is required for binding phosphatidylinositol 3-kinase. Journal of Biological Chemistry. 1996;271:26329–26334. - PubMed
-
- Ding Q, Grammer JR, Nelson MA, Guan JL, Stewart JE, Jr, Gladson CL. p27(Kip1) and cyclin D1 are necessary for focal adhesion kinase (FAK) regulation of cell cycle progression in glioblastoma cells propagated in vitro and in vivo in the scid mouse brain. Journal of Biological Chemistry. 2005;280:6802–6805. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

