Insulin signaling promotes germline proliferation in C. elegans
- PMID: 20110332
- PMCID: PMC2827619
- DOI: 10.1242/dev.042523
Insulin signaling promotes germline proliferation in C. elegans
Erratum in
- Development. 2014 Jan;141(1):237
Abstract
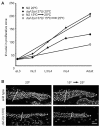
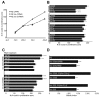
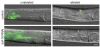
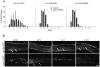
Cell proliferation must be coordinated with cell fate specification during development, yet interactions among pathways that control these two critical aspects of development are not well understood. The coordination of cell fate specification and proliferation is particularly crucial during early germline development, when it impacts the establishment of stem/progenitor cell populations and ultimately the production of gametes. In C. elegans, insulin/IGF-like receptor (IIR) signaling has been implicated in fertility, but the basis for the fertility defect had not been previously characterized. We found that IIR signaling is required for robust larval germline proliferation, separate from its well-characterized role in preventing dauer entry. IIR signaling stimulates the larval germline cell cycle. This activity is distinct from Notch signaling, occurs in a predominantly germline-autonomous manner, and responds to somatic activity of ins-3 and ins-33, genes that encode putative insulin-like ligands. IIR signaling in this role acts through the canonical PI3K pathway, inhibiting DAF-16/FOXO. However, signaling from these ligands does not inhibit daf-16 in neurons nor in the intestine, two tissues previously implicated in other IIR roles. Our data are consistent with a model in which: (1) under replete reproductive conditions, the larval germline responds to insulin signaling to ensure robust germline proliferation that builds up the germline stem cell population; and (2) distinct insulin-like ligands contribute to different phenotypes by acting on IIR signaling in different tissues.
Figures


Similar articles
-
Cell-nonautonomous signaling of FOXO/DAF-16 to the stem cells of Caenorhabditis elegans.PLoS Genet. 2012;8(8):e1002836. doi: 10.1371/journal.pgen.1002836. Epub 2012 Aug 16. PLoS Genet. 2012. PMID: 22916022 Free PMC article.
-
Control of cell-fate plasticity and maintenance of multipotency by DAF-16/FoxO in quiescent Caenorhabditis elegans.Proc Natl Acad Sci U S A. 2013 Feb 5;110(6):2181-6. doi: 10.1073/pnas.1222377110. Epub 2013 Jan 22. Proc Natl Acad Sci U S A. 2013. PMID: 23341633 Free PMC article.
-
Insulin signaling activity in the somatic gonad controls dauer entry via the germ line in C. elegans.Curr Biol. 2025 Aug 18;35(16):3986-3993.e4. doi: 10.1016/j.cub.2025.05.028. Epub 2025 Jul 16. Curr Biol. 2025. PMID: 40675151
-
Insulin and germline proliferation in Caenorhabditis elegans.Vitam Horm. 2011;87:61-77. doi: 10.1016/B978-0-12-386015-6.00024-X. Vitam Horm. 2011. PMID: 22127237 Free PMC article. Review.
-
The search for DAF-16/FOXO transcriptional targets: approaches and discoveries.Exp Gerontol. 2006 Oct;41(10):910-21. doi: 10.1016/j.exger.2006.06.040. Epub 2006 Aug 24. Exp Gerontol. 2006. PMID: 16934425 Review.
Cited by
-
Cell Cycle Analysis in the C. elegans Germline with the Thymidine Analog EdU.J Vis Exp. 2018 Oct 22;(140):58339. doi: 10.3791/58339. J Vis Exp. 2018. PMID: 30394383 Free PMC article.
-
IGF signalling in germ cells and testicular germ cell tumours: roles and therapeutic approaches.Andrology. 2019 Jul;7(4):536-544. doi: 10.1111/andr.12658. Epub 2019 Jun 9. Andrology. 2019. PMID: 31179642 Free PMC article. Review.
-
TATN-1 mutations reveal a novel role for tyrosine as a metabolic signal that influences developmental decisions and longevity in Caenorhabditis elegans.PLoS Genet. 2013;9(12):e1004020. doi: 10.1371/journal.pgen.1004020. Epub 2013 Dec 19. PLoS Genet. 2013. PMID: 24385923 Free PMC article.
-
Irises: A practical tool for image-based analysis of cellular DNA content.Worm. 2014 May 8;3:e29041. doi: 10.4161/worm.29041. eCollection 2014. Worm. 2014. PMID: 25254149 Free PMC article.
-
Autophagy in C. elegans development.Dev Biol. 2019 Mar 1;447(1):103-125. doi: 10.1016/j.ydbio.2018.04.009. Epub 2018 Apr 27. Dev Biol. 2019. PMID: 29709599 Free PMC article. Review.
References
-
- Apfeld J., Kenyon C. (1998). Cell nonautonomy of C. elegans daf-2 function in the regulation of diapause and life span. Cell 95, 199-210 - PubMed
-
- Austin J., Kimble J. (1987). glp-1 is required in the germ line for regulation of the decision between mitosis and meiosis in C. elegans. Cell 51, 589-599 - PubMed
-
- Bathgate R. A., Ivell R., Sanborn B. M., Sherwood O. D., Summers R. J. (2005). Receptors for relaxin family peptides. Ann. N Y Acad. Sci. 1041, 61-76 - PubMed
-
- Baumeister R., Schaffitzel E., Hertweck M. (2006). Endocrine signaling in Caenorhabditis elegans controls stress response and longevity. J. Endocrinol. 190, 191-202 - PubMed
-
- Berry L. W., Westlund B., Schedl T. (1997). Germ-line tumor formation caused by activation of glp-1, a Caenorhabditis elegans member of the Notch family of receptors. Development 124, 925-936 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

