Alternatively activated macrophages and collagen remodeling characterize the postpartum involuting mammary gland across species
- PMID: 20110414
- PMCID: PMC2832146
- DOI: 10.2353/ajpath.2010.090735
Alternatively activated macrophages and collagen remodeling characterize the postpartum involuting mammary gland across species
Abstract
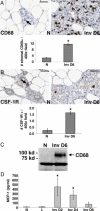
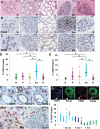
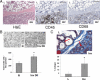
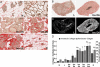
Recent pregnancy correlates with decreased survival for breast cancer patients compared with non-pregnancy-associated breast cancer. We hypothesize that postpartum mammary involution induces metastasis through wound-healing programs known to promote cancer. It is unknown whether alternatively activated M2 macrophages, immune cells important in wound-healing and experimental tumorigenesis that also predict poor prognosis for breast cancer patients, are recruited to the normal involuting gland. Macrophage markers CD68, CSF-1R, and F4/80 were examined across the pregnancy and involution cycle in rodent and human mammary tissues. Quantitative immunohistochemistry revealed up to an eightfold increase in macrophage number during involution, which returned to nulliparous levels with full regression. The involution macrophages exhibit an M2 phenotype as determined by high arginase-1 and low inducible nitric oxide synthase staining in rodent tissue, and by mannose receptor expression in human breast tissue. M2 cytokines IL-4 and IL-13 also peaked during involution. Extracellular matrix (ECM) isolated from involuting rat mammary glands was chemotactic for macrophages compared with nulliparous mammary ECM. Fibrillar collagen levels and proteolysis increased dramatically during involution, and denatured collagen I acted as a strong chemoattractant for macrophages in cell culture, suggesting proteolyzed fibrillar collagen as a candidate ECM mediator of macrophage recruitment. M2 macrophages, IL-4, IL-13, fibrillar collagen accumulation, and proteolysis of collagen are all components of tumor promotional microenvironments, and thus may mediate promotion of breast cancers arising in the postpartum setting.
Figures

Similar articles
-
Macphatics and PoEMs in Postpartum Mammary Development and Tumor Progression.J Mammary Gland Biol Neoplasia. 2020 Jun;25(2):103-113. doi: 10.1007/s10911-020-09451-6. Epub 2020 Jun 13. J Mammary Gland Biol Neoplasia. 2020. PMID: 32535810 Free PMC article. Review.
-
Non-steroidal anti-inflammatory drugs target the pro-tumorigenic extracellular matrix of the postpartum mammary gland.Int J Dev Biol. 2011;55(7-9):745-55. doi: 10.1387/ijdb.113379jo. Int J Dev Biol. 2011. PMID: 22161831
-
Wound healing-like immune program facilitates postpartum mammary gland involution and tumor progression.Int J Cancer. 2015 Apr 15;136(8):1803-13. doi: 10.1002/ijc.29181. Epub 2014 Sep 15. Int J Cancer. 2015. PMID: 25187059 Free PMC article.
-
Rat mammary extracellular matrix composition and response to ibuprofen treatment during postpartum involution by differential GeLC-MS/MS analysis.J Proteome Res. 2012 Oct 5;11(10):4894-905. doi: 10.1021/pr3003744. Epub 2012 Aug 30. J Proteome Res. 2012. PMID: 22897585 Free PMC article.
-
Mammary gland involution as an immunotherapeutic target for postpartum breast cancer.J Mammary Gland Biol Neoplasia. 2014 Jul;19(2):213-28. doi: 10.1007/s10911-014-9322-z. Epub 2014 Jun 22. J Mammary Gland Biol Neoplasia. 2014. PMID: 24952477 Free PMC article. Review.
Cited by
-
An "elite hacker": breast tumors exploit the normal microenvironment program to instruct their progression and biological diversity.Cell Adh Migr. 2012 May-Jun;6(3):236-48. doi: 10.4161/cam.20880. Epub 2012 May 1. Cell Adh Migr. 2012. PMID: 22863741 Free PMC article. Review.
-
Wounds that will not heal: pervasive cellular reprogramming in cancer.Am J Pathol. 2013 Apr;182(4):1055-64. doi: 10.1016/j.ajpath.2013.01.009. Epub 2013 Feb 22. Am J Pathol. 2013. PMID: 23438473 Free PMC article. Review.
-
Endometriosis, a disease of the macrophage.Front Immunol. 2013 Jan 28;4:9. doi: 10.3389/fimmu.2013.00009. eCollection 2013. Front Immunol. 2013. PMID: 23372570 Free PMC article.
-
Weighing the Risk: effects of Obesity on the Mammary Gland and Breast Cancer Risk.J Mammary Gland Biol Neoplasia. 2020 Jun;25(2):115-131. doi: 10.1007/s10911-020-09452-5. Epub 2020 Jun 9. J Mammary Gland Biol Neoplasia. 2020. PMID: 32519090 Free PMC article. Review.
-
Macphatics and PoEMs in Postpartum Mammary Development and Tumor Progression.J Mammary Gland Biol Neoplasia. 2020 Jun;25(2):103-113. doi: 10.1007/s10911-020-09451-6. Epub 2020 Jun 13. J Mammary Gland Biol Neoplasia. 2020. PMID: 32535810 Free PMC article. Review.
References
-
- Lambe M, Hsieh C, Trichopoulos D, Ekbom A, Pavia M, Adami HO. Transient increase in the risk of breast cancer after giving birth. N Engl J Med. 1994;331:5–9. - PubMed
-
- Chie WC, Hsieh C, Newcomb PA, Longnecker MP, Mittendorf R, Greenberg ER, Clapp RW, Burke KP, Titus-Ernstoff L, Trentham-Dietz A, MacMahon B. Age at any full-term pregnancy and breast cancer risk. Am J Epidemiol. 2000;151:715–722. - PubMed
-
- Liu Q, Wuu J, Lambe M, Hsieh SF, Ekbom A, Hsieh CC. Transient increase in breast cancer risk after giving birth: postpartum period with the highest risk (Sweden) Cancer Causes Control. 2002;13:299–305. - PubMed
-
- Schedin P. Pregnancy-associated breast cancer and metastasis. Nat Rev Cancer. 2006;6:281–291. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

