Plant antimicrobial agents and their effects on plant and human pathogens
- PMID: 20111686
- PMCID: PMC2812829
- DOI: 10.3390/ijms10083400
Plant antimicrobial agents and their effects on plant and human pathogens
Abstract
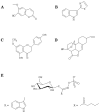
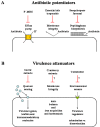
To protect themselves, plants accumulate an armoury of antimicrobial secondary metabolites. Some metabolites represent constitutive chemical barriers to microbial attack (phytoanticipins) and others inducible antimicrobials (phytoalexins). They are extensively studied as promising plant and human disease-controlling agents. This review discusses the bioactivity of several phytoalexins and phytoanticipins defending plants against fungal and bacterial aggressors and those with antibacterial activities against pathogens affecting humans such as Pseudomonas aeruginosa and Staphylococcus aureus involved in respiratory infections of cystic fibrosis patients. The utility of plant products as "antibiotic potentiators" and "virulence attenuators" is also described as well as some biotechnological applications in phytoprotection.
Keywords: antimicrobial; cystic fibrosis; infectious disease; phytoalexin; phytoanticipin; phytoprotection; secondary metabolites.
Figures

Similar articles
-
Chemical diversity and defence metabolism: how plants cope with pathogens and ozone pollution.Int J Mol Sci. 2009 Jul 30;10(8):3371-3399. doi: 10.3390/ijms10083371. Int J Mol Sci. 2009. PMID: 20111684 Free PMC article. Review.
-
Phytoalexins in defense against pathogens.Trends Plant Sci. 2012 Feb;17(2):73-90. doi: 10.1016/j.tplants.2011.11.002. Epub 2011 Dec 30. Trends Plant Sci. 2012. PMID: 22209038 Review.
-
Fungal resistance to plant antibiotics as a mechanism of pathogenesis.Microbiol Mol Biol Rev. 1999 Sep;63(3):708-24. doi: 10.1128/MMBR.63.3.708-724.1999. Microbiol Mol Biol Rev. 1999. PMID: 10477313 Free PMC article. Review.
-
Antimicrobial and anti-Quorum Sensing activities of selected medicinal plants of Ethiopia: Implication for development of potent antimicrobial agents.BMC Microbiol. 2016 Jul 11;16(1):139. doi: 10.1186/s12866-016-0765-9. BMC Microbiol. 2016. PMID: 27400878 Free PMC article.
-
Effect of Bacoside A on growth and biofilm formation by Staphylococcus aureus and Pseudomonas aeruginosa.Can J Microbiol. 2017 Feb;63(2):169-178. doi: 10.1139/cjm-2016-0365. Epub 2016 Oct 24. Can J Microbiol. 2017. PMID: 28099040
Cited by
-
Dose-Dependent Alterations to In Vitro Human Microbiota Composition and Butyrate Inhibition by a Supercritical Carbon Dioxide Hops Extract.Biomolecules. 2019 Aug 21;9(9):390. doi: 10.3390/biom9090390. Biomolecules. 2019. PMID: 31438572 Free PMC article.
-
Wound-Healing and Antibacterial Activity of the Quercetin-4-Formyl Phenyl Boronic Acid Complex against Bacterial Pathogens of Diabetic Foot Ulcer.ACS Omega. 2022 Jul 8;7(28):24415-24422. doi: 10.1021/acsomega.2c01819. eCollection 2022 Jul 19. ACS Omega. 2022. PMID: 35874257 Free PMC article.
-
Disease Resistance Mechanisms in Plants.Genes (Basel). 2018 Jul 4;9(7):339. doi: 10.3390/genes9070339. Genes (Basel). 2018. PMID: 29973557 Free PMC article. Review.
-
Botrytis cinerea manipulates the antagonistic effects between immune pathways to promote disease development in tomato.Plant Cell. 2011 Jun;23(6):2405-21. doi: 10.1105/tpc.111.083394. Epub 2011 Jun 10. Plant Cell. 2011. PMID: 21665999 Free PMC article.
-
Genomic basis of broad host range and environmental adaptability of Rhizobium tropici CIAT 899 and Rhizobium sp. PRF 81 which are used in inoculants for common bean (Phaseolus vulgaris L.).BMC Genomics. 2012 Dec 27;13:735. doi: 10.1186/1471-2164-13-735. BMC Genomics. 2012. PMID: 23270491 Free PMC article.
References
-
- Bais HP, Weir TL, Perry LG, Gilroy S, Vivanco JM. The role of root exudates in rhizosphere interactions with plants and other organisms. Annu. Rev. Plant Biol. 2006;57:233–266. - PubMed
-
- Grayer RJ, Kokubun T. Plant-fungal interactions: The search for phytoalexins and other antifungal compounds from higher plants. Phytochemistry. 2001;56:253–263. - PubMed
-
- Jones JD, Dangl JL. The plant immune system. Nature. 2006;444:323–329. - PubMed
-
- Lindsay WP, Lamb CJ, Dixon RA. Microbial recognition and activation of plant defense systems. Trends Microbiol. 1993;1:181–187. - PubMed
-
- Bednarek P, Osbourn A. Plant-microbe interactions: Chemical diversity in plant defense. Science. 2009;324:746–748. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

