Activation-induced T helper cell death contributes to Th1/Th2 polarization following murine Schistosoma japonicum infection
- PMID: 20111738
- PMCID: PMC2810471
- DOI: 10.1155/2010/202397
Activation-induced T helper cell death contributes to Th1/Th2 polarization following murine Schistosoma japonicum infection
Abstract
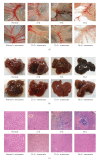
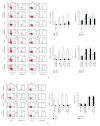
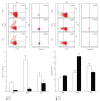
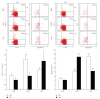
In chronic infectious diseases, such as schistosomiasis, pathogen growth and immunopathology are affected by the induction of a proper balanced Th1/Th2 response to the pathogen and by antigen-triggered activation-induced T cell death. Here, by using S. japonicum infection or schistosome antigens-immunized mouse model, or antigens in vitro stimulation, we report that during the early stage of S. japonicum infection, nonegg antigens trigger Th2 cell apoptosis via the granzyme B signal pathway, contributing to Th1 polarization, which is thought to be associated with worm clearance and severe schistosomiasis. Meanwhile, after the adult worms lay their eggs, the egg antigens trigger Th1 cell apoptosis via the caspase pathway, contributing to Th2 polarization, which is associated with mild pathology and enhanced survival of both worms and their hosts. Thus, our study suggests that S. japonicum antigen-induced Th1 and Th2 cell apoptosis involves the Th1/Th2 shift and favorites both hosts and parasites.
Figures


Similar articles
-
T cell-mediated immunity in CBA mice during Schistosoma japonicum infection.Exp Parasitol. 2019 Sep;204:107725. doi: 10.1016/j.exppara.2019.107725. Epub 2019 Jul 12. Exp Parasitol. 2019. PMID: 31306646
-
[Effects of soluble adult worm antigen and soluble egg antigen of Schistosoma japonicum on differentiation of CD4+ T cells of mice].Zhongguo Xue Xi Chong Bing Fang Zhi Za Zhi. 2013 Apr;25(2):151-6. Zhongguo Xue Xi Chong Bing Fang Zhi Za Zhi. 2013. PMID: 23894835 Chinese.
-
Dynamic analysis of T-lymphocyte function in relation to hepatopathologic changes and effect of interleukin-12 treatment in mice infected with Schistosoma japonicum.J Parasitol. 1999 Apr;85(2):257-62. J Parasitol. 1999. PMID: 10219305
-
T Lymphocyte-Mediated Liver Immunopathology of Schistosomiasis.Front Immunol. 2020 Feb 18;11:61. doi: 10.3389/fimmu.2020.00061. eCollection 2020. Front Immunol. 2020. PMID: 32132991 Free PMC article. Review.
-
The immunobiology of Th1 polarization in high-pathology schistosomiasis.Immunol Rev. 2004 Oct;201:168-79. doi: 10.1111/j.0105-2896.2004.00197.x. Immunol Rev. 2004. PMID: 15361240 Review.
Cited by
-
Network model of immune responses reveals key effectors to single and co-infection dynamics by a respiratory bacterium and a gastrointestinal helminth.PLoS Comput Biol. 2012 Jan;8(1):e1002345. doi: 10.1371/journal.pcbi.1002345. Epub 2012 Jan 12. PLoS Comput Biol. 2012. PMID: 22253585 Free PMC article.
-
Excretory/Secretory Products from Schistosoma japonicum Eggs Alleviate Ovalbumin-Induced Allergic Airway Inflammation.PLoS Negl Trop Dis. 2023 Oct 3;17(10):e0011625. doi: 10.1371/journal.pntd.0011625. eCollection 2023 Oct. PLoS Negl Trop Dis. 2023. PMID: 37788409 Free PMC article.
-
The dynamic changes of CD3e-CD11c+ dendritic cells in spleens and bone marrow of mice infected with Schistosoma japonicum.Parasitol Res. 2017 Mar;116(3):1007-1011. doi: 10.1007/s00436-017-5381-6. Epub 2017 Feb 10. Parasitol Res. 2017. PMID: 28185057
-
CD4+ Th2 cells are directly regulated by IL-10 during allergic airway inflammation.Mucosal Immunol. 2017 Jan;10(1):150-161. doi: 10.1038/mi.2016.47. Epub 2016 May 11. Mucosal Immunol. 2017. PMID: 27166557
-
Changes in NK and NKT cells in mesenteric lymph nodes after a Schistosoma japonicum infection.Parasitol Res. 2014 Mar;113(3):1001-9. doi: 10.1007/s00436-013-3732-5. Epub 2013 Dec 10. Parasitol Res. 2014. PMID: 24322293
References
-
- Infante-Duarte C. Th1/Th2 balance in infection. Springer Seminars in Immunopathology. 1999;21(3):317–338. - PubMed
-
- Rogers PR, Croft M. Peptide dose, affinity, and time of differentiation can contribute to the Th1/Th2 cytokine balance. Journal of Immunology. 1999;163(3):1205–1213. - PubMed
-
- Balkhi MY, Latchumanan VK, Singh B, Sharma P, Natarajan K. Cross-regulation of CD86 by CD80 differentially regulates T helper responses from Mycobacterium tuberculosis secretory antigen-activated dendritic cell subsets. Journal of Leukocyte Biology. 2004;75(5):874–883. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

