Nicotine facilitates long-term potentiation induction in oriens-lacunosum moleculare cells via Ca2+ entry through non-alpha7 nicotinic acetylcholine receptors
- PMID: 20113344
- PMCID: PMC2852628
- DOI: 10.1111/j.1460-9568.2009.07058.x
Nicotine facilitates long-term potentiation induction in oriens-lacunosum moleculare cells via Ca2+ entry through non-alpha7 nicotinic acetylcholine receptors
Abstract
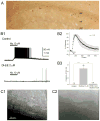
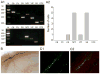
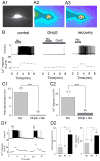
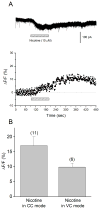
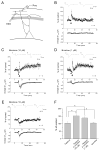
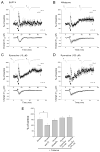
Hippocampal inhibitory interneurons have a central role in the control of network activity, and excitatory synapses that they receive express Hebbian and anti-Hebbian long-term potentiation (LTP). Because many interneurons in the hippocampus express nicotinic acetylcholine receptors (nAChRs), we explored whether exposure to nicotine promotes LTP induction in these interneurons. We focussed on a subset of interneurons in the stratum oriens/alveus that were continuously activated in the presence of nicotine due to the expression of non-desensitizing non-alpha7 nAChRs. We found that, in addition to alpha2 subunit mRNAs, these interneurons were consistently positive for somatostatin and neuropeptide Y mRNAs, and showed morphological characteristics of oriens-lacunosum moleculare cells. Activation of non-alpha7 nAChRs increased intracellular Ca(2+) levels at least in part via Ca(2+) entry through their channels. Presynaptic tetanic stimulation induced N-methyl-D-aspartate receptor-independent LTP in voltage-clamped interneurons at -70 mV when in the presence, but not absence, of nicotine. Intracellular application of a Ca(2+) chelator blocked LTP induction, suggesting the requirement of Ca(2+) signal for LTP induction. The induction of LTP was still observed in the presence of ryanodine, which inhibits Ca(2+) -induced Ca(2+) release from ryanodine-sensitive intracellular stores, and the L-type Ca(2+) channel blocker nifedipine. These results suggest that Ca(2+) entry through non-alpha7 nAChR channels is critical for LTP induction. Thus, nicotine affects hippocampal network activity by promoting LTP induction in oriens-lacunosum moleculare cells via continuous activation of non-alpha7 nAChRs.
Figures
References
-
- Abdulla FA, Bradbury E, Calaminici MR, Lippiello PM, Wonnacott S, Gray JA, Sinden JD. Relationship between up-regulation of nicotine binding sites in rat brain and delayed cognitive enhancement observed after chronic or acute nicotinic receptor stimulation. Psychopharmacology. 1996;124:323–331. - PubMed
-
- Alkondon M, Pereira EF, Almeida LE, Randall WR, Albuquerque EX. Nicotine at concentrations found in cigarette smokers activates and desensitizes nicotinic acetylcholine receptors in CA1 interneurons of rat hippocampus. Neuropharmacology. 2000;39:2726–2739. - PubMed
-
- Alkondon M, Albuquerque EX. Nicotinic acetylcholine receptor alpha7 and alpha4beta2 subtypes differentially control GABAergic input to CA1 neurons in rat hippocampus. J Neurophysiol. 2001;86:3043–55. - PubMed
-
- Alkondon M, Albuquerque EX. Nicotinic receptor subtypes in rat hippocampal slices are differentially sensitive to desensitization and early in vivo functional up-regulation by nicotine and to block by bupropion. J Pharmacol Exp Ther. 2005;313:740–750. - PubMed
-
- Benowitz NL, Porchet H, Jacob P. Nicotine dependence and tolerance in man: pharmacokinetic and pharmacodynamic investigations. Prog Brain Res. 1989;79:279–287. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

