Dissecting the roles of DR4, DR5 and c-FLIP in the regulation of geranylgeranyltransferase I inhibition-mediated augmentation of TRAIL-induced apoptosis
- PMID: 20113484
- PMCID: PMC2824632
- DOI: 10.1186/1476-4598-9-23
Dissecting the roles of DR4, DR5 and c-FLIP in the regulation of geranylgeranyltransferase I inhibition-mediated augmentation of TRAIL-induced apoptosis
Abstract
Background: Geranylgeranyltransferase I (GGTase I) has emerged as a cancer therapeutic target. Accordingly, small molecules that inhibit GGTase I have been developed and exhibit encouraging anticancer activity in preclinical studies. However, their underlying anticancer mechanisms remain unclear. Here we have demonstrated a novel mechanism by which GGTase I inhibition modulates apoptosis.
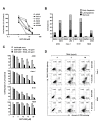
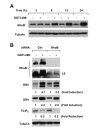
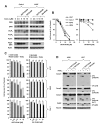
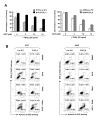
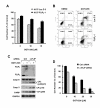
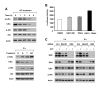
Results: The GGTase I inhibitor GGTI-298 induced apoptosis and augmented tumor necrosis factor-related apoptosis-inducing ligand (TRAIL)-induced apoptosis in human lung cancer cells. GGTI-298 induced DR4 and DR5 expression and reduced c-FLIP levels. Enforced c-FLIP expression or DR5 knockdown attenuated apoptosis induced by GGTI-298 and TRAIL combination. Surprisingly, DR4 knockdown sensitized cancer cells to GGTI298/TRAIL-induced apoptosis. The combination of GGTI-298 and TRAIL was more effective than each single agent in decreasing the levels of IkappaBalpha and p-Akt, implying that GGTI298/TRAIL activates NF-kappaB and inhibits Akt. Interestingly, knockdown of DR5, but not DR4, prevented GGTI298/TRAIL-induced IkappaBalpha and p-Akt reduction, suggesting that DR5 mediates reduction of IkappaBalpha and p-Akt induced by GGTI298/TRAIL. In contrast, DR4 knockdown further facilitated GGTI298/TRAIL-induced p-Akt reduction.
Conclusions: Both DR5 induction and c-FLIP downregulation contribute to GGTI-298-mediated augmentation of TRAIL-induced apoptosis. Moreover, DR4 appears to play an opposite role to DR5 in regulation of GGTI/TRAIL-induced apoptotic signaling.
Figures


Similar articles
-
Suppression of HSP70 expression sensitizes NSCLC cell lines to TRAIL-induced apoptosis by upregulating DR4 and DR5 and downregulating c-FLIP-L expressions.J Mol Med (Berl). 2013 Feb;91(2):219-35. doi: 10.1007/s00109-012-0947-3. Epub 2012 Sep 5. J Mol Med (Berl). 2013. PMID: 22948392
-
The eIF4E/eIF4G interaction inhibitor 4EGI-1 augments TRAIL-mediated apoptosis through c-FLIP Down-regulation and DR5 induction independent of inhibition of cap-dependent protein translation.Neoplasia. 2010 Apr;12(4):346-56. doi: 10.1593/neo.10144. Neoplasia. 2010. PMID: 20360945 Free PMC article.
-
The alkylphospholipid perifosine induces apoptosis of human lung cancer cells requiring inhibition of Akt and activation of the extrinsic apoptotic pathway.Mol Cancer Ther. 2007 Jul;6(7):2029-38. doi: 10.1158/1535-7163.MCT-07-0004. Epub 2007 Jun 29. Mol Cancer Ther. 2007. PMID: 17604333
-
Down-regulation of intracellular anti-apoptotic proteins, particularly c-FLIP by therapeutic agents; the novel view to overcome resistance to TRAIL.J Cell Physiol. 2018 Oct;233(10):6470-6485. doi: 10.1002/jcp.26585. Epub 2018 May 9. J Cell Physiol. 2018. PMID: 29741767 Review.
-
c-FLIP, a master anti-apoptotic regulator.Exp Oncol. 2012 Oct;34(3):176-84. Exp Oncol. 2012. PMID: 23070002 Free PMC article. Review.
Cited by
-
Andrographolide Enhances TRAIL-Induced Apoptosis via p53-Mediated Death Receptors Up-Regulation and Suppression of the NF-кB Pathway in Bladder Cancer Cells.Int J Biol Sci. 2019 Jan 24;15(3):688-700. doi: 10.7150/ijbs.30847. eCollection 2019. Int J Biol Sci. 2019. PMID: 30745855 Free PMC article.
-
YM155 sensitizes HeLa cells to TRAIL-mediated apoptosis via cFLIP and survivin downregulation.Oncol Lett. 2020 Oct;20(4):72. doi: 10.3892/ol.2020.11933. Epub 2020 Jul 30. Oncol Lett. 2020. PMID: 32863905 Free PMC article.
-
Cytoplasmic levels of cFLIP determine a broad susceptibility of breast cancer stem/progenitor-like cells to TRAIL.Mol Cancer. 2015 Dec 15;14:209. doi: 10.1186/s12943-015-0478-y. Mol Cancer. 2015. PMID: 26667821 Free PMC article.
-
Targeting apoptosis to manage acquired resistance to third generation EGFR inhibitors.Front Med. 2022 Oct;16(5):701-713. doi: 10.1007/s11684-022-0951-0. Epub 2022 Sep 24. Front Med. 2022. PMID: 36152124 Review.
-
Induction of IL6/STAT3-dependent TRAIL expression that contributes to the therapeutic efficacy of osimertinib in EGFR mutant NSCLC cells.Oncogene. 2025 Jul;44(27):2315-2327. doi: 10.1038/s41388-025-03381-5. Epub 2025 Apr 17. Oncogene. 2025. PMID: 40247082
References
-
- Wagner KW, Punnoose EA, Januario T, Lawrence DA, Pitti RM, Lancaster K, Lee D, von Goetz M, Yee SF, Totpal K, Huw L, Katta V, Cavet G, Hymowitz SG, Amler L, Ashkenazi A. Death-receptor O-glycosylation controls tumor-cell sensitivity to the proapoptotic ligand Apo2L/TRAIL. Nat Med. 2007;13:1070–1077. doi: 10.1038/nm1627. - DOI - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

