Methylation of novel markers of fragile X alleles is inversely correlated with FMRP expression and FMR1 activation ratio
- PMID: 20118148
- PMCID: PMC2846165
- DOI: 10.1093/hmg/ddq037
Methylation of novel markers of fragile X alleles is inversely correlated with FMRP expression and FMR1 activation ratio
Abstract
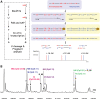
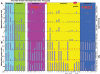
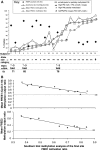
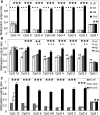
The fragile X syndrome (FXS) is caused by silencing of the fragile X mental retardation gene (FMR1) and the absence of its product, fragile X mental retardation protein (FMRP), resulting from CpG island methylation associated with large CGG repeat expansions (more than 200) termed full mutation (FM). We have identified a number of novel epigenetic markers for FXS using matrix-assisted laser desorption/ionization-time of flight mass spectrometry (MALDI-TOF MS), naming the most informative fragile X-related epigenetic element 1 (FREE1) and 2 (FREE2). Methylation of both regions was correlated with that of the FMR1 CpG island detected using Southern blot (FREE1 R = 0.97; P < 0.00001, n = 23 and FREE2 R = 0.93; P < 0.00001, n = 23) and negatively correlated with lymphocyte expression of FMRP (FREE1 R = -0.62; P = 0.01, n = 15 and FREE2 R = -0.55; P = 0.03, n = 15) in blood of partially methylated 'high functioning' FM males. In blood of FM carrier females, methylation of both markers was inversely correlated with the FMR1 activation ratio (FREE1 R = -0.93; P < 0.0001, n = 12 and FREE2 R = -0.95; P < 0.0001, n = 9). In a sample set of 49 controls, 18 grey zone (GZ 40-54 repeats), 22 premutation (PM 55-170 repeats) and 22 (affected) FXS subjects, the FREE1 methylation pattern was consistent between blood and chorionic villi as a marker of methylated FM alleles and could be used to differentiate FXS males and females from controls, as well as from carriers of GZ/PM alleles, but not between GZ and PM alleles and controls. Considering its high-throughput and specificity for pathogenic FM alleles, low cost and minimal DNA requirements, FREE MALDI-TOF MS offers a unique tool in FXS diagnostics and newborn population screening.
Figures

References
-
- Verkerk A.J., Pieretti M., Sutcliffe J.S., Fu Y.H., Kuhl D.P., Pizzuti A., Reiner O., Richards S., Victoria M.F., Zhang F.P., et al. Identification of a gene (FMR-1) containing a CGG repeat coincident with a breakpoint cluster region exhibiting length variation in fragile X syndrome. Cell. 1991;65:905–914. - PubMed
-
- Pieretti M., Zhang F.P., Fu Y.H., Warren S.T., Oostra B.A., Caskey C.T., Nelson D.L. Absence of expression of the FMR-1 gene in fragile X syndrome. Cell. 1991;66:817–822. - PubMed
-
- Irwin S.A., Galvez R., Greenough W.T. Dendritic spine structural anomalies in fragile-X mental retardation syndrome. Cereb. Cortex. 2000;10:1038–1044. - PubMed
-
- Jin P., Warren S.T. Understanding the molecular basis of fragile X syndrome. Hum. Mol. Genet. 2000;9:901–908. - PubMed
-
- Hagerman R.J., Leehey M., Heinrichs W., Tassone F., Wilson R., Hills J., Grigsby J., Gage B., Hagerman P.J. Intention tremor, parkinsonism, and generalized brain atrophy in male carriers of fragile X. Neurology. 2001;57:127–130. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

