Airway epithelial indoleamine 2,3-dioxygenase inhibits CD4+ T cells during Aspergillus fumigatus antigen exposure
- PMID: 20118221
- PMCID: PMC3028254
- DOI: 10.1165/rcmb.2009-0167OC
Airway epithelial indoleamine 2,3-dioxygenase inhibits CD4+ T cells during Aspergillus fumigatus antigen exposure
Abstract
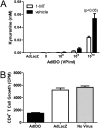
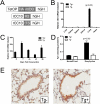
Indoleamine 2,3-dioxygenase (IDO) suppresses the functions of CD4(+) T cells through its ability to metabolize the essential amino acid tryptophan. Although the activity of IDO is required for the immunosuppression of allergic airway disease by the Toll-Like-Receptor 9 (TLR9) agonist, oligonucleotides comprised of cytosine and guanine nucleotides linked by phosphodiester bonds (CpG) DNA, it is unclear whether IDO expression by resident lung epithelial cells is sufficient to elicit these effects. Therefore, we created a transgenic mouse inducibly overexpressing IDO within nonciliated airway epithelial cells. Upon inhalation of formalin-fixed Aspergillus fumigatus hyphal antigens, the overexpression of IDO from airway epithelial cells of these mice reduced the number of CD4(+) T cells within the inflamed lung and impaired the capacity of antigen-specific splenic CD4(+) effector T cells to secrete the cytokines IL-4, IL-5, IL-13, and IFN-γ. Despite these effects, allergic airway disease pathology was largely unaffected in mice expressing IDO in airway epithelium. In support of the concept that dendritic cells are the major cell type contributing to the IDO-inducing effects of CpG DNA, mice expressing TLR9 only in the airway epithelium did not augment IDO expression subsequent to the administration of CpG DNA. Furthermore, the systemic depletion of CD11c(+) cells rendered mice incapable of CpG DNA-induced IDO expression. Our results demonstrate that an overexpression of IDO within the airway epithelium represents a novel mechanism by which the number of CD4(+) T cells recruited to the lung and their capacity to produce cytokines can be diminished in a model of allergic airway disease, and these results also highlight the critical role of dendritic cells in the antiasthmatic effects of IDO induction by CpG DNA.
Figures
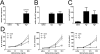
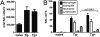



Similar articles
-
Non-hematopoietic cells contribute to protective tolerance to Aspergillus fumigatus via a TRIF pathway converging on IDO.Cell Mol Immunol. 2010 Nov;7(6):459-70. doi: 10.1038/cmi.2010.43. Epub 2010 Sep 13. Cell Mol Immunol. 2010. PMID: 20835271 Free PMC article.
-
Immature dendritic cells expressing indoleamine 2,3-dioxygenase suppress ovalbumin-induced allergic airway inflammation in mice.J Investig Allergol Clin Immunol. 2011;21(3):185-92. J Investig Allergol Clin Immunol. 2011. PMID: 21548446
-
IDO expressing fibroblasts promote the expansion of antigen specific regulatory T cells.Immunobiology. 2014 Jan;219(1):17-24. doi: 10.1016/j.imbio.2013.06.008. Epub 2013 Jun 25. Immunobiology. 2014. PMID: 23891282
-
HIV inhibits CD4+ T-cell proliferation by inducing indoleamine 2,3-dioxygenase in plasmacytoid dendritic cells.Blood. 2007 Apr 15;109(8):3351-9. doi: 10.1182/blood-2006-07-034785. Epub 2006 Dec 7. Blood. 2007. PMID: 17158233 Free PMC article. Clinical Trial.
-
IDO: a double-edged sword for T(H)1/T(H)2 regulation.Immunol Lett. 2008 Nov 16;121(1):1-6. doi: 10.1016/j.imlet.2008.08.008. Epub 2008 Sep 29. Immunol Lett. 2008. PMID: 18824197 Free PMC article. Review.
Cited by
-
Weight Loss Decreases Inherent and Allergic Methacholine Hyperresponsiveness in Mouse Models of Diet-Induced Obese Asthma.Am J Respir Cell Mol Biol. 2016 Aug;55(2):176-87. doi: 10.1165/rcmb.2016-0070OC. Am J Respir Cell Mol Biol. 2016. PMID: 27064658 Free PMC article.
-
Targeting immunometabolism in host-directed therapies to fungal disease.Clin Exp Immunol. 2022 Jun 11;208(2):158-166. doi: 10.1093/cei/uxab014. Clin Exp Immunol. 2022. PMID: 35641161 Free PMC article. Review.
-
Dissecting the inflammatory twitch in allergically inflamed mice.Am J Physiol Lung Cell Mol Physiol. 2016 May 15;310(10):L1003-9. doi: 10.1152/ajplung.00036.2016. Epub 2016 Mar 4. Am J Physiol Lung Cell Mol Physiol. 2016. PMID: 26944087 Free PMC article.
-
Non-hematopoietic cells contribute to protective tolerance to Aspergillus fumigatus via a TRIF pathway converging on IDO.Cell Mol Immunol. 2010 Nov;7(6):459-70. doi: 10.1038/cmi.2010.43. Epub 2010 Sep 13. Cell Mol Immunol. 2010. PMID: 20835271 Free PMC article.
-
Mesenchymal stromal cells mediate Aspergillus hyphal extract-induced allergic airway inflammation by inhibition of the Th17 signaling pathway.Stem Cells Transl Med. 2014 Feb;3(2):194-205. doi: 10.5966/sctm.2013-0061. Epub 2014 Jan 16. Stem Cells Transl Med. 2014. PMID: 24436442 Free PMC article.
References
-
- Wang JM, Rambaldi A, Biondi A, Chen ZG, Sanderson CJ, Mantovani A. Recombinant human interleukin 5 is a selective eosinophil chemoattractant. Eur J Immunol 1989;19:701–705. - PubMed
-
- Yang M, Hogan SP, Henry PJ, Matthaei KI, McKenzie AN, Young IG, Rothenberg ME, Foster PS. Interleukin-13 mediates airways hyperreactivity through the IL-4 receptor-alpha chain and STAT-6 independently of IL-5 and eotaxin. Am J Respir Cell Mol Biol 2001;25:522–530. - PubMed
-
- Coffman RL, Ohara J, Bond MW, Carty J, Zlotnik A, Paul WE. B cell stimulatory factor-1 enhances the IgE response of lipopolysaccharide-activated B cells. J Immunol 1986;136:4538–4541. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

