Insights into the effect of soil pH on N(2)O and N(2) emissions and denitrifier community size and activity
- PMID: 20118356
- PMCID: PMC2838029
- DOI: 10.1128/AEM.02484-09
Insights into the effect of soil pH on N(2)O and N(2) emissions and denitrifier community size and activity
Abstract
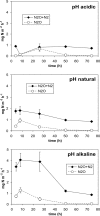
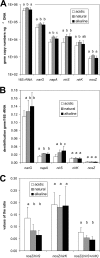
The objective of this study was to investigate how changes in soil pH affect the N(2)O and N(2) emissions, denitrification activity, and size of a denitrifier community. We established a field experiment, situated in a grassland area, which consisted of three treatments which were repeatedly amended with a KOH solution (alkaline soil), an H(2)SO(4) solution (acidic soil), or water (natural pH soil) over 10 months. At the site, we determined field N(2)O and N(2) emissions using the (15)N gas flux method and collected soil samples for the measurement of potential denitrification activity and quantification of the size of the denitrifying community by quantitative PCR of the narG, napA, nirS, nirK, and nosZ denitrification genes. Overall, our results indicate that soil pH is of importance in determining the nature of denitrification end products. Thus, we found that the N(2)O/(N(2)O + N(2)) ratio increased with decreasing pH due to changes in the total denitrification activity, while no changes in N(2)O production were observed. Denitrification activity and N(2)O emissions measured under laboratory conditions were correlated with N fluxes in situ and therefore reflected treatment differences in the field. The size of the denitrifying community was uncoupled from in situ N fluxes, but potential denitrification was correlated with the count of NirS denitrifiers. Significant relationships were observed between nirS, napA, and narG gene copy numbers and the N(2)O/(N(2)O + N(2)) ratio, which are difficult to explain. However, this highlights the need for further studies combining analysis of denitrifier ecology and quantification of denitrification end products for a comprehensive understanding of the regulation of N fluxes by denitrification.
Figures

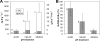
References
-
- Adams, T. M., and S. N. Adams. 1983. The effects of liming and soil pH on carbon and nitrogen contained in the soil biomass. J. Agric. Sci. 101:553-558.
-
- Arah, J. R. M. 1997. Apportioning nitrous oxide fluxes between nitrification and denitrification using gas-phase mass spectrometry. Soil Biol. Biochem. 29:1295-1299.
-
- Baudoin, E., L. Philippot, D. Chèneby, L. Chapuis-Lardy, N. Fromin, D. Bru, B. Rabary, and A. Brauman. 2009. Direct seeding mulch-based cropping increases both the activity and abundance of denitrifier communities in a tropical soil. Soil Biol. Biochem. 41:1703-1709.
-
- Cavigelli, M. A., and G. P. Robertson. 2000. The functional significance of denitrifier community composition in a terrestrial ecosystem. Ecology 81:1402-1414.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

