Insights into prion protein function from atomistic simulations
- PMID: 20118658
- PMCID: PMC2850415
- DOI: 10.4161/pri.4.1.10969
Insights into prion protein function from atomistic simulations
Abstract
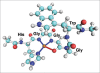
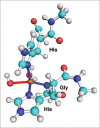
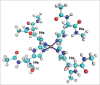
Computer simulations are a powerful tool for studies of biological systems. They have often been used to study prion protein (PrP), a protein responsible for neurodegenerative diseases, which include "mad cow disease" in cattle and Creutzfeldt-Jacob disease in humans. An important aspect of the prion protein is its interaction with copper ion, which is thought to be relevant for PrP's yet undetermined function and also potentially play a role in prion diseases. for studies of copper attachment to the prion protein, computer simulations have often been used to complement experimental data and to obtain binding structures of Cu-PrP complexes. This paper summarizes the results of recent ab initio calculations of copper-prion protein interactions focusing on the recently discovered concentration-dependent binding modes in the octarepeat region of this protein. In addition to determining the binding structures, computer simulations were also used to make predictions about PrP's function and the role of copper in prion diseases. The results demonstrate the predictive power and applicability of ab initio simulations for studies of metal-biomolecular complexes.
Figures
References
-
- Prusiner SB. Prion Diseases and the BSE Crisis. Science. 1997;278:245–251. - PubMed
-
- Baxa U, Cassese T, Kajava AV, Steven AC. Structure, function and amyloidogenesis of fungal prions: filament polymorphism and prion variants. Adv Protein Chem. 2006;73:125–180. - PubMed
-
- Klamt F, Dal-Pizzol F, Conte da Frota ML, Walz R, Andrades ME, da Silva EG, et al. Imbalance of antioxidant defense in mice lacking cellular prion protein. Free Radic Biol Med. 2001;30:1137–1144. - PubMed
-
- Vassallo N, Herms J. Cellular prion protein function in copper homeostasis and redox signalling at the synapse. J Neurochem. 2003;86:538–544. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
