Contribution of each of four Superantigens to Streptococcus equi-induced mitogenicity, gamma interferon synthesis, and immunity
- PMID: 20123710
- PMCID: PMC2849420
- DOI: 10.1128/IAI.01079-09
Contribution of each of four Superantigens to Streptococcus equi-induced mitogenicity, gamma interferon synthesis, and immunity
Abstract
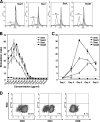
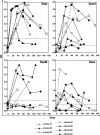
Streptococcus equi is the causative agent of strangles, the most frequently diagnosed infectious disease of horses worldwide. The disease is characterized by abscessation and swelling of the lymph nodes of the head and neck, which can literally strangle the horse to death. S. equi produces four recently acquired phage-associated bacterial superantigens (sAgs; SeeH, SeeI, SeeL, and SeeM) that share homology with the mitogenic toxins of Streptococcus pyogenes. The aim of this study was to characterize the contribution of each of these S. equi sAgs to mitogenic activity in vitro and quantify the sAg-neutralizing capacity of sera from naturally infected horses in order to better understand their role in pathogenicity. Each of the sAgs was successfully cloned, and soluble proteins were produced in Escherichia coli. SeeI, SeeL, and SeeM induced a dose-dependent proliferative response in equine CD4 T lymphocytes and synthesis of gamma interferon (IFN-gamma). SeeH did not stimulate equine peripheral blood mononuclear cells (PBMC) but induced proliferation of asinine PBMC. Allelic replacement mutants of S. equi strain 4047 with sequential deletion of the superantigen genes were generated. Deletion of seeI, seeL, and seeM completely abrogated the mitogenic activity and synthesis of IFN-gamma, in equine PBMC, of the strain 4047 culture supernatant. Sera from naturally infected convalescent horses had only limited sAg-neutralizing activities. We propose that S. equi sAgs play an important role in S. equi pathogenicity by stimulating an overzealous and inappropriate Th1 response that may interfere with the development of an effective immune response.
Figures




Similar articles
-
Novel Streptococcus equi strains causing strangles outbreaks in Arabian horses in Egypt.Transbound Emerg Dis. 2020 Nov;67(6):2455-2466. doi: 10.1111/tbed.13584. Epub 2020 May 10. Transbound Emerg Dis. 2020. PMID: 32304282
-
Identification of three novel superantigen-encoding genes in Streptococcus equi subsp. zooepidemicus, szeF, szeN, and szeP.Infect Immun. 2010 Nov;78(11):4817-27. doi: 10.1128/IAI.00751-10. Epub 2010 Aug 16. Infect Immun. 2010. PMID: 20713629 Free PMC article.
-
Localised mitogenic activity in horses following infection with Streptococcus equi.Res Vet Sci. 2015 Jun;100:100-4. doi: 10.1016/j.rvsc.2015.03.015. Epub 2015 Mar 11. Res Vet Sci. 2015. PMID: 25841794
-
Streptococcus zooepidemicus and Streptococcus equi evolution: the role of CRISPRs.Biochem Soc Trans. 2013 Dec;41(6):1437-43. doi: 10.1042/BST20130165. Biochem Soc Trans. 2013. PMID: 24256234 Review.
-
New perspectives for the diagnosis, control, treatment, and prevention of strangles in horses.Vet Clin North Am Equine Pract. 2014 Dec;30(3):591-607. doi: 10.1016/j.cveq.2014.08.007. Epub 2014 Oct 6. Vet Clin North Am Equine Pract. 2014. PMID: 25300634 Review.
Cited by
-
Genetic analysis based on next generation sequencing of Streptococcus equi subsp. equi isolated from horses imported into Japan.J Vet Med Sci. 2024 Aug 2;86(8):828-832. doi: 10.1292/jvms.23-0342. Epub 2024 Jun 18. J Vet Med Sci. 2024. PMID: 38897953 Free PMC article.
-
Streptococcal sagA activates a proinflammatory response in mast cells by a sublytic mechanism.Cell Microbiol. 2019 Sep;21(9):e13064. doi: 10.1111/cmi.13064. Epub 2019 Jul 7. Cell Microbiol. 2019. PMID: 31155820 Free PMC article.
-
Transmission of Streptococcus equi subspecies zooepidemicus infection from horses to humans.Emerg Infect Dis. 2013 Jul;19(7):1041-8. doi: 10.3201/eid1907.121365. Emerg Infect Dis. 2013. PMID: 23777752 Free PMC article.
-
Differences in the Accessory Genomes and Methylomes of Strains of Streptococcus equi subsp. equi and of Streptococcus equi subsp. zooepidemicus Obtained from the Respiratory Tract of Horses from Texas.Microbiol Spectr. 2022 Feb 23;10(1):e0076421. doi: 10.1128/spectrum.00764-21. Epub 2022 Jan 12. Microbiol Spectr. 2022. PMID: 35019696 Free PMC article.
-
Genomic characterization of Streptococcus equi subspecies zooepidemicus from a 2021 outbreak in Indiana with increased sow mortality.mSphere. 2023 Dec 20;8(6):e0040423. doi: 10.1128/msphere.00404-23. Epub 2023 Oct 20. mSphere. 2023. PMID: 37861318 Free PMC article.
References
-
- Alber, J., A. El-Sayed, S. Estoepangestie, C. Lammler, and M. Zschock. 2005. Dissemination of the superantigen encoding genes seeL, seeM, szeL and szeM in Streptococcus equi subsp. equi and Streptococcus equi subsp. zooepidemicus. Vet. Microbiol. 109:135-141. - PubMed
-
- Anzai, T., A. S. Sheoran, Y. Kuwamoto, T. Kondo, R. Wada, T. Inoue, and J. F. Timoney. 1999. Streptococcus equi but not Streptococcus zooepidemicus produces potent mitogenic responses from equine peripheral blood mononuclear cells. Vet. Immunol. Immunopathol. 67:235-246. - PubMed
-
- Arad, G., D. Hillman, R. Levy, and R. Kaempfer. 2004. Broad-spectrum immunity against superantigens is elicited in mice protected from lethal shock by a superantigen antagonist peptide. Immunol. Lett. 91:141-145. - PubMed
-
- Arad, G., D. Hillman, R. Levy, and R. Kaempfer. 2001. Superantigen antagonist blocks Th1 cytokine gene induction and lethal shock. J. Leukoc. Biol. 69:921-927. - PubMed
-
- Artiushin, S. C., J. F. Timoney, A. S. Sheoran, and S. K. Muthupalani. 2002. Characterization and immunogenicity of pyrogenic mitogens SePE-H and SePE-I of Streptococcus equi. Microb. Pathog. 32:71-85. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

