Endogenous 3,4-dihydroxyphenylalanine and dopaquinone modifications on protein tyrosine: links to mitochondrially derived oxidative stress via hydroxyl radical
- PMID: 20124354
- PMCID: PMC2877980
- DOI: 10.1074/mcp.M900321-MCP200
Endogenous 3,4-dihydroxyphenylalanine and dopaquinone modifications on protein tyrosine: links to mitochondrially derived oxidative stress via hydroxyl radical
Abstract
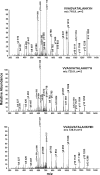
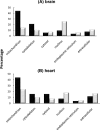
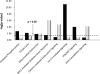
Oxidative modifications of protein tyrosines have been implicated in multiple human diseases. Among these modifications, elevations in levels of 3,4-dihydroxyphenylalanine (DOPA), a major product of hydroxyl radical addition to tyrosine, has been observed in a number of pathologies. Here we report the first proteome survey of endogenous site-specific modifications, i.e. DOPA and its further oxidation product dopaquinone in mouse brain and heart tissues. Results from LC-MS/MS analyses included 50 and 14 DOPA-modified tyrosine sites identified from brain and heart, respectively, whereas only a few nitrotyrosine-containing peptides, a more commonly studied marker of oxidative stress, were detectable, suggesting the much higher abundance for DOPA modification as compared with tyrosine nitration. Moreover, 20 and 12 dopaquinone-modified peptides were observed from brain and heart, respectively; nearly one-fourth of these peptides were also observed with DOPA modification on the same sites. For both tissues, these modifications are preferentially found in mitochondrial proteins with metal binding properties, consistent with metal-catalyzed hydroxyl radical formation from mitochondrial superoxide and hydrogen peroxide. These modifications also link to a number of mitochondrially associated and other signaling pathways. Furthermore, many of the modification sites were common sites of previously reported tyrosine phosphorylation, suggesting potential disruption of signaling pathways. Collectively, the results suggest that these modifications are linked with mitochondrially derived oxidative stress and may serve as sensitive markers for disease pathologies.
Figures
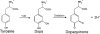

Similar articles
-
Evidence for covalent lipoyl adduction with dopaquinone following tyrosinase-catalyzed oxidation.Biochem Biophys Res Commun. 2006 Apr 28;343(1):15-20. doi: 10.1016/j.bbrc.2006.02.118. Epub 2006 Feb 28. Biochem Biophys Res Commun. 2006. PMID: 16527255
-
Quantum chemical approach to the mechanism for the biological conversion of tyrosine to dopaquinone.J Am Chem Soc. 2008 Dec 17;130(50):16890-7. doi: 10.1021/ja802618s. J Am Chem Soc. 2008. PMID: 19007228
-
The effects of hydroxyl radical attack on dopa, dopamine, 6-hydroxydopa, and 6-hydroxydopamine.Pigment Cell Res. 1995 Dec;8(6):283-93. doi: 10.1111/j.1600-0749.1995.tb00676.x. Pigment Cell Res. 1995. PMID: 8789736
-
Mitochondrial protein tyrosine nitration.Free Radic Res. 2011 Jan;45(1):37-52. doi: 10.3109/10715762.2010.516254. Epub 2010 Oct 13. Free Radic Res. 2011. PMID: 20942571 Review.
-
Chemistry of mixed melanogenesis--pivotal roles of dopaquinone.Photochem Photobiol. 2008 May-Jun;84(3):582-92. doi: 10.1111/j.1751-1097.2007.00238.x. Photochem Photobiol. 2008. PMID: 18435614 Review.
Cited by
-
Parkinson's Disease and Melanoma: Co-Occurrence and Mechanisms.J Parkinsons Dis. 2018;8(3):385-398. doi: 10.3233/JPD-171263. J Parkinsons Dis. 2018. PMID: 29991141 Free PMC article. Review.
-
Proteomic approaches to analyze protein tyrosine nitration.Antioxid Redox Signal. 2013 Oct 10;19(11):1247-56. doi: 10.1089/ars.2012.5058. Epub 2013 Jan 3. Antioxid Redox Signal. 2013. PMID: 23157221 Free PMC article. Review.
-
Tyrosine modifications in aging.Antioxid Redox Signal. 2012 Dec 1;17(11):1571-9. doi: 10.1089/ars.2012.4595. Epub 2012 May 14. Antioxid Redox Signal. 2012. PMID: 22424390 Free PMC article. Review.
-
Enzymatic Phosphorylation of Oxidized Tyrosine Residues.J Proteome Res. 2023 Jun 2;22(6):1959-1968. doi: 10.1021/acs.jproteome.3c00061. Epub 2023 May 5. J Proteome Res. 2023. PMID: 37146082 Free PMC article.
-
Ablation of Sam50 is associated with fragmentation and alterations in metabolism in murine and human myotubes.bioRxiv [Preprint]. 2023 Oct 19:2023.05.20.541602. doi: 10.1101/2023.05.20.541602. bioRxiv. 2023. PMID: 37292887 Free PMC article. Preprint.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

