A mouse model of severe halothane hepatitis based on human risk factors
- PMID: 20124411
- PMCID: PMC2872951
- DOI: 10.1124/jpet.109.164541
A mouse model of severe halothane hepatitis based on human risk factors
Abstract
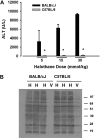
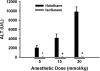
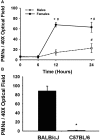
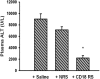
Halothane (2-bromo-2-chloro-1,1,1-trifluoro-ethane) is an inhaled anesthetic that induces severe, idiosyncratic liver injury, i.e., "halothane hepatitis," in approximately 1 in 20,000 human patients. We used known human risk factors (female sex, adult age, and genetics) as well as probable risk factors (fasting and inflammatory stress) to develop a murine model with characteristics of human halothane hepatitis. Female and male BALB/cJ mice treated with halothane developed dose-dependent liver injury within 24 h; however, the liver injury was severe only in females. Livers had extensive centrilobular necrosis, inflammatory cell infiltrate, and steatosis. Fasting rendered mice more sensitive to halothane hepatotoxicity, and 8-week-old female mice were more sensitive than males of the same age or than younger (4-week-old) females. C57BL/6 mice were insensitive to halothane, suggesting a strong genetic predisposition. In halothane-treated females, plasma concentration of tumor necrosis factor-alpha was greater than in males, and neutrophils were recruited to liver more rapidly and to a greater extent. Anti-CD18 serum attenuated halothane-induced liver injury in female mice, suggesting that neutrophil migration, activation, or both are required for injury. Coexposure of halothane-treated male mice to lipopolysaccharide to induce modest inflammatory stress converted their mild hepatotoxic response to a pronounced, female-like response. This is the first animal model of an idiosyncratic adverse drug reaction that is based on human risk factors and produces reproducible, severe hepatitis from halothane exposure with lesions characteristic of human halothane hepatitis. Moreover, these results suggest that a more robust innate immune response underlies the predisposition of female mice to halothane hepatitis.
Figures








Similar articles
-
Natural killer cells mediate severe liver injury in a murine model of halothane hepatitis.Toxicol Sci. 2011 Apr;120(2):507-18. doi: 10.1093/toxsci/kfr005. Epub 2011 Jan 18. Toxicol Sci. 2011. PMID: 21245496 Free PMC article.
-
Role of neutrophils in a mouse model of halothane-induced liver injury.Hepatology. 2006 Dec;44(6):1421-31. doi: 10.1002/hep.21425. Hepatology. 2006. PMID: 17133481
-
Lipopolysaccharide and trovafloxacin coexposure in mice causes idiosyncrasy-like liver injury dependent on tumor necrosis factor-alpha.Toxicol Sci. 2007 Nov;100(1):259-66. doi: 10.1093/toxsci/kfm218. Epub 2007 Aug 19. Toxicol Sci. 2007. PMID: 17709330
-
Isoflurane hepatotoxicity: a case report and review of the literature.Am J Gastroenterol. 1996 Nov;91(11):2406-9. Am J Gastroenterol. 1996. PMID: 8931426 Review.
-
Halothane hepatitis.Br J Anaesth. 1991 Jul;67(1):84-99. doi: 10.1093/bja/67.1.84. Br J Anaesth. 1991. PMID: 1859766 Review.
Cited by
-
Animal models of drug-induced liver injury.Biochim Biophys Acta Mol Basis Dis. 2019 May 1;1865(5):1031-1039. doi: 10.1016/j.bbadis.2018.08.037. Epub 2018 Sep 3. Biochim Biophys Acta Mol Basis Dis. 2019. PMID: 31007174 Free PMC article. Review.
-
Drug-Induced Liver Injury: Cascade of Events Leading to Cell Death, Apoptosis or Necrosis.Int J Mol Sci. 2017 May 9;18(5):1018. doi: 10.3390/ijms18051018. Int J Mol Sci. 2017. PMID: 28486401 Free PMC article. Review.
-
Immune Modulation by Volatile Anesthetics.Anesthesiology. 2016 Aug;125(2):399-411. doi: 10.1097/ALN.0000000000001195. Anesthesiology. 2016. PMID: 27286478 Free PMC article. Review.
-
The hepatotoxicity of Polygonum multiflorum: The emerging role of the immune-mediated liver injury.Acta Pharmacol Sin. 2021 Jan;42(1):27-35. doi: 10.1038/s41401-020-0360-3. Epub 2020 Mar 2. Acta Pharmacol Sin. 2021. PMID: 32123300 Free PMC article. Review.
-
Roles of Cofactors in Drug-Induced Liver Injury: Drug Metabolism and Beyond.Drug Metab Dispos. 2022 May;50(5):646-654. doi: 10.1124/dmd.121.000457. Epub 2022 Feb 27. Drug Metab Dispos. 2022. PMID: 35221288 Free PMC article. Review.
References
-
- Becker PU, Deis A, Sørensen TI, Grønbaek MN, Borch-Johnsen K, Müller CF, Schnohr P, Jensen GB. (1997) [Alcohol intake and risk of liver disease–significance of gender. A population study]. Ugeskr Laeger 159:3782–3786 - PubMed
-
- Berger D, Bölke E, Seidelmann M, Beger HG. (1997) Time-scale of interleukin-6, myeloid related proteins (MRP), C reactive protein (CRP), and endotoxin plasma levels during the postoperative acute phase reaction. Shock 7:422–426 - PubMed
-
- Bird GL, Williams R. (1989) Detection of antibodies to a halothane metabolite hepten in sera from patients with halothane-associated hepatitis. J Hepatol 9:366–373 - PubMed
-
- Blackburn WR, Ngai SH, Lindenbaum J. (1964) Morphologic changes in hepatic necrosis following halothane anesthesia in man. Anesthesiology 25:270–283 - PubMed
-
- Bourdi M, Amouzadeh HR, Rushmore TH, Martin JL, Pohl LR. (2001) Halothane-induced liver injury in outbred guinea pigs: role of trifluoroacetylated protein adducts in animal susceptibility. Chem Res Toxicol 14:362–370 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

