Expression map of the human exome in CD34+ cells and blood cells: increased alternative splicing in cell motility and immune response genes
- PMID: 20126548
- PMCID: PMC2813875
- DOI: 10.1371/journal.pone.0008990
Expression map of the human exome in CD34+ cells and blood cells: increased alternative splicing in cell motility and immune response genes
Abstract
Background: Hematopoietic cells are endowed with very specific biological functions, including cell motility and immune response. These specific functions are dramatically altered during hematopoietic cell differentiation, whereby undifferentiated hematopoietic stem and progenitor cells (HSPC) residing in bone marrow differentiate into platelets, red blood cells and immune cells that exit into the blood stream and eventually move into lymphoid organs or inflamed tissues. The contribution of alternative splicing (AS) to these functions has long been minimized due to incomplete knowledge on AS events in hematopoietic cells.
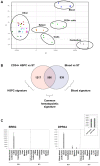
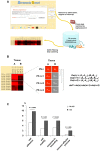
Principal findings: Using Human Exon ST 1.0 microarrays, the entire exome expression profile of immature CD34+ HSPC and mature whole blood cells was mapped, compared to a collection of solid tissues and made freely available as an online exome expression atlas (Amazonia Exon! : http://amazonia.transcriptome.eu/exon.php). At a whole transcript level, HSPC strongly expressed EREG and the pluripotency marker DPPA4. Using a differential splicing index scheme (dsi), a list of 849 transcripts differentially expressed between hematopoietic cells and solid tissues was computed, that included NEDD9 and CD74. Some of these genes also underwent alternative splicing events during hematopoietic differentiation, such as INPP4B, PTPLA or COMMD6, with varied contribution of CD3+ T cells, CD19+ B cells, CD14+ or CD15+ myelomonocytic populations. Strikingly, these genes were significantly enriched for genes involved in cell motility, cell adhesion, response to wounding and immune processes.
Conclusion: The relevance and the precision provided by this exon expression map highlights the contribution of alternative splicing to key feature of blood cells differentiation and function.
Conflict of interest statement
Figures


Similar articles
-
Transcriptome profiling and sequencing of differentiated human hematopoietic stem cells reveal lineage-specific expression and alternative splicing of genes.Physiol Genomics. 2011 Oct 20;43(20):1117-34. doi: 10.1152/physiolgenomics.00099.2011. Epub 2011 Aug 9. Physiol Genomics. 2011. PMID: 21828245 Free PMC article.
-
Microarray and serial analysis of gene expression analyses identify known and novel transcripts overexpressed in hematopoietic stem cells.Cancer Res. 2004 Jul 1;64(13):4434-41. doi: 10.1158/0008-5472.CAN-03-3247. Cancer Res. 2004. PMID: 15231652
-
Differential gene expression underlying the functional distinctions of primary human CD34+ hematopoietic stem and progenitor cells from peripheral blood and bone marrow.Ann N Y Acad Sci. 2003 May;996:89-100. doi: 10.1111/j.1749-6632.2003.tb03237.x. Ann N Y Acad Sci. 2003. PMID: 12799287
-
Alternative splicing and differential gene expression in colon cancer detected by a whole genome exon array.BMC Genomics. 2006 Dec 27;7:325. doi: 10.1186/1471-2164-7-325. BMC Genomics. 2006. PMID: 17192196 Free PMC article.
-
Gene expression profiling identifies significant differences between the molecular phenotypes of bone marrow-derived and circulating human CD34+ hematopoietic stem cells.Blood. 2002 Mar 15;99(6):2037-44. doi: 10.1182/blood.v99.6.2037. Blood. 2002. PMID: 11877277
Cited by
-
Problems with using the normal distribution--and ways to improve quality and efficiency of data analysis.PLoS One. 2011;6(7):e21403. doi: 10.1371/journal.pone.0021403. Epub 2011 Jul 14. PLoS One. 2011. PMID: 21779325 Free PMC article.
-
Stacking the DEK: from chromatin topology to cancer stem cells.Cell Cycle. 2013 Jan 1;12(1):51-66. doi: 10.4161/cc.23121. Epub 2012 Dec 19. Cell Cycle. 2013. PMID: 23255114 Free PMC article. Review.
-
TET2 mutations improve the new European LeukemiaNet risk classification of acute myeloid leukemia: a Cancer and Leukemia Group B study.J Clin Oncol. 2011 Apr 1;29(10):1373-81. doi: 10.1200/JCO.2010.32.7742. Epub 2011 Feb 22. J Clin Oncol. 2011. PMID: 21343549 Free PMC article. Clinical Trial.
-
Analysis of the transcriptome in molecular epidemiology studies.Environ Mol Mutagen. 2013 Aug;54(7):500-17. doi: 10.1002/em.21798. Epub 2013 Aug 1. Environ Mol Mutagen. 2013. PMID: 23907930 Free PMC article. Review.
-
Inositol polyphosphate-4-phosphatase type II plays critical roles in the modulation of cadherin-mediated adhesion dynamics of pancreatic ductal adenocarcinomas.Cell Adh Migr. 2018;12(6):548-563. doi: 10.1080/19336918.2018.1491496. Epub 2018 Aug 19. Cell Adh Migr. 2018. PMID: 29952716 Free PMC article.
References
Publication types
MeSH terms
Substances
Associated data
- Actions
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Research Materials

