A novel molecule integrating therapeutic and diagnostic activities reveals multiple aspects of stem cell-based therapy
- PMID: 20127797
- PMCID: PMC3021283
- DOI: 10.1002/stem.313
A novel molecule integrating therapeutic and diagnostic activities reveals multiple aspects of stem cell-based therapy
Abstract
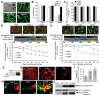
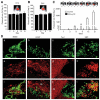
Stem cells are promising therapeutic delivery vehicles; however pre-clinical and clinical applications of stem cell-based therapy would benefit significantly from the ability to simultaneously determine therapeutic efficacy and pharmacokinetics of therapies delivered by engineered stem cells. In this study, we engineered and screened numerous fusion variants that contained therapeutic (TRAIL) and diagnostic (luciferase) domains designed to allow simultaneous investigation of multiple events in stem cell-based therapy in vivo. When various stem cell lines were engineered with the optimized molecule, SRL(O)L(2)TR, diagnostic imaging showed marked differences in the levels and duration of secretion between stem cell lines, while the therapeutic activity of the molecule showed the different secretion levels translated to significant variability in tumor cell killing. In vivo, simultaneous diagnostic and therapeutic monitoring revealed that stem cell-based delivery significantly improved pharmacokinetics and anti-tumor effectiveness of the therapy compared to intravenous or intratumoral delivery. As treatment for highly malignant brain tumor xenografts, tracking SRL(O)L(2)TR showed stable stem cell-mediated delivery significantly regressed peripheral and intracranial tumors. Together, the integrated diagnostic and therapeutic properties of SRL(O)L(2)TR answer critical questions necessary for successful utilization of stem cells as novel therapeutic vehicles.
Figures



Similar articles
-
A first-generation multi-functional cytokine for simultaneous optical tracking and tumor therapy.PLoS One. 2012;7(7):e40234. doi: 10.1371/journal.pone.0040234. Epub 2012 Jul 11. PLoS One. 2012. PMID: 22808125 Free PMC article.
-
Nanoparticle engineered TRAIL-overexpressing adipose-derived stem cells target and eradicate glioblastoma via intracranial delivery.Proc Natl Acad Sci U S A. 2016 Nov 29;113(48):13857-13862. doi: 10.1073/pnas.1615396113. Epub 2016 Nov 14. Proc Natl Acad Sci U S A. 2016. PMID: 27849590 Free PMC article.
-
Tumoricidal stem cell therapy enables killing in novel hybrid models of heterogeneous glioblastoma.Neuro Oncol. 2019 Dec 17;21(12):1552-1564. doi: 10.1093/neuonc/noz138. Neuro Oncol. 2019. PMID: 31420675 Free PMC article.
-
Stem cell-based therapies for tumors in the brain: are we there yet?Neuro Oncol. 2016 Aug;18(8):1066-78. doi: 10.1093/neuonc/now096. Epub 2016 Jun 9. Neuro Oncol. 2016. PMID: 27282399 Free PMC article. Review.
-
Encapsulated stem cells for cancer therapy.Biomatter. 2013 Jan-Mar;3(1):e24278. doi: 10.4161/biom.24278. Epub 2013 Jan 1. Biomatter. 2013. PMID: 23507920 Free PMC article. Review.
Cited by
-
Mesenchymal Stem Cell-Based COVID-19 Therapy: Bioengineering Perspectives.Cells. 2022 Jan 29;11(3):465. doi: 10.3390/cells11030465. Cells. 2022. PMID: 35159275 Free PMC article. Review.
-
Mesenchymal stem cells engineered for cancer therapy.Adv Drug Deliv Rev. 2012 Jun 1;64(8):739-48. doi: 10.1016/j.addr.2011.06.010. Epub 2011 Jun 29. Adv Drug Deliv Rev. 2012. PMID: 21740940 Free PMC article. Review.
-
Electrospun nanofibrous scaffolds increase the efficacy of stem cell-mediated therapy of surgically resected glioblastoma.Biomaterials. 2016 Jun;90:116-25. doi: 10.1016/j.biomaterials.2016.03.008. Epub 2016 Mar 9. Biomaterials. 2016. PMID: 27016620 Free PMC article.
-
Bi-specific molecule against EGFR and death receptors simultaneously targets proliferation and death pathways in tumors.Sci Rep. 2017 Jun 1;7(1):2602. doi: 10.1038/s41598-017-02483-9. Sci Rep. 2017. PMID: 28572590 Free PMC article.
-
Real-time multi-modality imaging of glioblastoma tumor resection and recurrence.J Neurooncol. 2013 Jan;111(2):153-61. doi: 10.1007/s11060-012-1008-z. Epub 2012 Dec 16. J Neurooncol. 2013. PMID: 23242736 Free PMC article.
References
-
- Corsten MF, Shah K. Therapeutic stem-cells for cancer treatment: Hopes and hurdles in tactical warfare. Lancet Oncology. 2008;9:376–384. - PubMed
-
- Daley GQ, Scadden DT. Prospects for stem cell-based therapy. Cell. 2008;132:544–548. - PubMed
-
- Corsten MF, Miranda R, Kasmieh R, et al. MicroRNA-21 knockdown disrupts glioma growth in vivo and displays synergistic cytotoxicity with neural precursor cell delivered S-TRAIL in human gliomas. Cancer Research. 2007;67:8994–9000. - PubMed
-
- Yip S, Sabetrasekh R, Sidman RL, et al. Neural stem cells as novel cancer therapeutic vehicles. Eur J Cancer. 2006;42:1298–1308. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

