The fibroblast integrin alpha11beta1 is induced in a mechanosensitive manner involving activin A and regulates myofibroblast differentiation
- PMID: 20129924
- PMCID: PMC2856250
- DOI: 10.1074/jbc.M109.078766
The fibroblast integrin alpha11beta1 is induced in a mechanosensitive manner involving activin A and regulates myofibroblast differentiation
Abstract
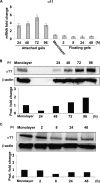
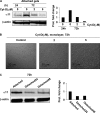
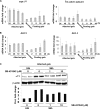
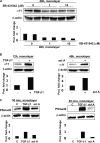
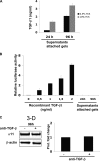
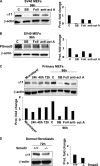
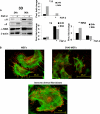
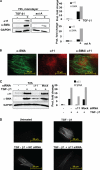
Fibrotic tissue is characterized by an overabundance of myofibroblasts. Thus, understanding the factors that induce myofibroblast differentiation is paramount to preventing fibrotic healing. Previous studies have shown that mechanical stress derived from the integrin-mediated interaction between extracellular matrix and the cytoskeleton promotes myofibroblast differentiation. Integrin alpha11beta1 is a collagen receptor on fibroblasts. To determine whether alpha11beta1 can act as a mechanosensor to promote the myofibroblast phenotype, mouse embryonic fibroblasts and human corneal fibroblasts were utilized. We found that alpha11 mRNA and protein levels were up-regulated in mouse embryonic fibroblasts grown in attached three-dimensional collagen gels and conversely down-regulated in cells grown in floating gels. alpha11 up-regulation could be prevented by manually detaching the collagen gels or by cytochalasin D treatment. Furthermore, SB-431542, an inhibitor of signaling via ALK4, ALK5, and ALK7, prevented the up-regulation of alpha11 and the concomitant phosphorylation of Smad3 under attached conditions. In attached gels, TGF-beta1 was secreted in its inactive form but surprisingly not further activated, thus not influencing alpha11 regulation. However, inhibition of activin A attenuated the up-regulation of alpha11. To determine the role of alpha11 in myofibroblast differentiation, human corneal fibroblasts were transfected with small interfering RNA to alpha11, which decreased alpha-smooth muscle actin expression and myofibroblast differentiation. Our data suggest that alpha11beta1 is regulated by cell/matrix stress involving activin A and Smad3 and that alpha11beta1 regulates myofibroblast differentiation.
Figures
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

