Optimized adeno-associated viral vector-mediated striatal DOPA delivery restores sensorimotor function and prevents dyskinesias in a model of advanced Parkinson's disease
- PMID: 20129936
- PMCID: PMC2822630
- DOI: 10.1093/brain/awp314
Optimized adeno-associated viral vector-mediated striatal DOPA delivery restores sensorimotor function and prevents dyskinesias in a model of advanced Parkinson's disease
Abstract
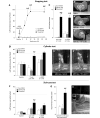
Viral vector-mediated gene transfer utilizing adeno-associated viral vectors has recently entered clinical testing as a novel tool for delivery of therapeutic agents to the brain. Clinical trials in Parkinson's disease using adeno-associated viral vector-based gene therapy have shown the safety of the approach. Further efforts in this area will show if gene-based approaches can rival the therapeutic efficacy achieved with the best pharmacological therapy or other, already established, surgical interventions. One of the strategies under development for clinical application is continuous 3,4-dihydroxyphenylalanine delivery. This approach has been shown to be efficient in restoring motor function and reducing established dyskinesias in rats with a partial lesion of the nigrostriatal dopamine projection. Here we utilized high purity recombinant adeno-associated viral vectors serotype 5 coding for tyrosine hydroxylase and its co-factor synthesizing enzyme guanosine-5'-triphosphate cyclohydrolase-1, delivered at an optimal ratio of 5 : 1, to show that the enhanced 3,4-dihydroxyphenylalanine production obtained with this optimized delivery system results in robust recovery of function in spontaneous motor tests after complete dopamine denervation. We found that the therapeutic efficacy was substantial and could be maintained for at least 6 months. The tyrosine hydroxylase plus guanosine-5'-triphosphate cyclohydrolase-1 treated animals were resistant to developing dyskinesias upon peripheral l-3,4-dihydroxyphenylalanine drug challenge, which is consistent with the interpretation that continuous dopamine stimulation resulted in a normalization of the post-synaptic response. Interestingly, recovery of forelimb use in the stepping test observed here was maintained even after a second lesion depleting the serotonin input to the forebrain, suggesting that the therapeutic efficacy was not solely dependent on dopamine synthesis and release from striatal serotonergic terminals. Taken together these results show that vector-mediated continuous 3,4-dihydroxyphenylalanine delivery has the potential to provide significant symptomatic relief even in advanced stages of Parkinson's disease.
Figures


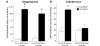
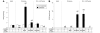

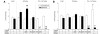
References
-
- Ahlenius S. Reversal by l-DOPA of the suppression of locomotor activity induced by inhibition of tyrosine-hydroxylase and DA-beta-hydroxylase in mice. Brain Res. 1974;69:57–65. - PubMed
-
- Ahlenius S, Andén NE, Engel J. Restoration of locomotor activity in mice by low l-DOPA doses after suppression by alpha-methyltyrosine but not by reserpine. Brain Res. 1973;62:189–99. - PubMed
-
- Arai R, Karasawa N, Geffard M, Nagatsu I. l-DOPA is converted to dopamine in serotonergic fibers of the striatum of the rat: a double-labeling immunofluorescence study. Neurosci Lett. 1995;195:195–8. - PubMed
-
- Arai R, Karasawa N, Geffard M, Nagatsu T, Nagatsu I. Immunohistochemical evidence that central serotonin neurons produce dopamine from exogenous l-DOPA in the rat, with reference to the involvement of aromatic L-amino acid decarboxylase. Brain Res. 1994;667:295–9. - PubMed
-
- Arai R, Karasawa N, Nagatsu I. Aromatic L-amino acid decarboxylase is present in serotonergic fibers of the striatum of the rat. A double-labeling immunofluorescence study. Brain Res. 1996;706:177–9. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

