The role of RPA2 phosphorylation in homologous recombination in response to replication arrest
- PMID: 20130019
- PMCID: PMC3916738
- DOI: 10.1093/carcin/bgq035
The role of RPA2 phosphorylation in homologous recombination in response to replication arrest
Abstract
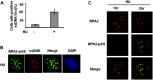
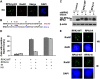
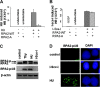
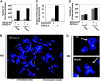
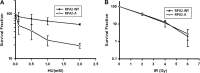
Failure to reactivate stalled or collapsed DNA replication forks is a potential source of genomic instability. Homologous recombination (HR) is a major mechanism for repairing the DNA damage resulting from replication arrest. The single-strand DNA (ssDNA)-binding protein, replication protein A (RPA), plays a major role in multiple processes of DNA metabolism. However, the role of RPA2 hyperphosphorylation, which occurs in response to DNA damage, had been unclear. Here, we show that hyperphosphorylated RPA2 associates with ssDNA and recombinase protein Rad51 in response to replication arrest by hydroxyurea (HU) treatment. In addition, RPA2 hyperphosphorylation is critical for Rad51 recruitment and HR-mediated repair following HU. However, RPA2 hyperphosphorylation is not essential for both ionizing radiation (IR)-induced Rad51 foci formation and I-Sce-I endonuclease-stimulated HR. Moreover, we show that expression of a phosphorylation-deficient mutant of RPA2 leads to increased chromosomal aberrations following HU treatment but not after exposure to IR. Finally, we demonstrate that loss of RPA2 hyperphosphorylation results in a loss of viability when cells are confronted with replication stress whereas cells expressing hyperphosphorylation-defective RPA2 or wild-type RPA2 have a similar sensitivity to IR. Thus, our data suggest that RPA2 hyperphosphorylation plays a critical role in maintenance of genomic stability and cell survival after a DNA replication block via promotion of HR.
Figures

Similar articles
-
Valproic acid sensitizes breast cancer cells to hydroxyurea through inhibiting RPA2 hyperphosphorylation-mediated DNA repair pathway.DNA Repair (Amst). 2017 Oct;58:1-12. doi: 10.1016/j.dnarep.2017.08.002. Epub 2017 Aug 9. DNA Repair (Amst). 2017. PMID: 28837865
-
Extensive RPA2 hyperphosphorylation promotes apoptosis in response to DNA replication stress in CHK1 inhibited cells.Nucleic Acids Res. 2015 Nov 16;43(20):9776-87. doi: 10.1093/nar/gkv835. Epub 2015 Aug 13. Nucleic Acids Res. 2015. PMID: 26271993 Free PMC article.
-
Differential involvement of phosphatidylinositol 3-kinase-related protein kinases in hyperphosphorylation of replication protein A2 in response to replication-mediated DNA double-strand breaks.Genes Cells. 2006 Mar;11(3):237-46. doi: 10.1111/j.1365-2443.2006.00942.x. Genes Cells. 2006. PMID: 16483312
-
Replication protein A, the laxative that keeps DNA regular: The importance of RPA phosphorylation in maintaining genome stability.Semin Cell Dev Biol. 2019 Feb;86:112-120. doi: 10.1016/j.semcdb.2018.04.005. Epub 2018 Nov 13. Semin Cell Dev Biol. 2019. PMID: 29665433 Review.
-
Moonlighting at replication forks - a new life for homologous recombination proteins BRCA1, BRCA2 and RAD51.FEBS Lett. 2017 Apr;591(8):1083-1100. doi: 10.1002/1873-3468.12556. Epub 2017 Jan 30. FEBS Lett. 2017. PMID: 28079255 Review.
Cited by
-
Inhibiting the DNA damage response as a therapeutic manoeuvre in cancer.Br J Pharmacol. 2013 Aug;169(8):1745-65. doi: 10.1111/bph.12244. Br J Pharmacol. 2013. PMID: 23682925 Free PMC article. Review.
-
p53 suppresses hyper-recombination by modulating BRCA1 function.DNA Repair (Amst). 2015 Sep;33:60-9. doi: 10.1016/j.dnarep.2015.06.005. Epub 2015 Jun 24. DNA Repair (Amst). 2015. PMID: 26162908 Free PMC article.
-
TTK inhibition radiosensitizes basal-like breast cancer through impaired homologous recombination.J Clin Invest. 2020 Feb 3;130(2):958-973. doi: 10.1172/JCI130435. J Clin Invest. 2020. PMID: 31961339 Free PMC article.
-
RFWD3-Dependent Ubiquitination of RPA Regulates Repair at Stalled Replication Forks.Mol Cell. 2015 Oct 15;60(2):280-93. doi: 10.1016/j.molcel.2015.09.011. Mol Cell. 2015. PMID: 26474068 Free PMC article.
-
Long non-coding RNA PCAT19 safeguards DNA in quiescent endothelial cells by preventing uncontrolled phosphorylation of RPA2.Cell Rep. 2022 Nov 15;41(7):111670. doi: 10.1016/j.celrep.2022.111670. Cell Rep. 2022. PMID: 36384122 Free PMC article.
References
-
- O'Driscoll M, et al. The role of double-strand break repair—insights from human genetics. Nat. Rev. Genet. 2006;7:45–54. - PubMed
-
- Lovett ST. Connecting replication and recombination. Mol. Cell. 2003;11:554–556. - PubMed
-
- Heller RC, et al. Replisome assembly and the direct restart of stalled replication forks. Nat. Rev. 2006;7:932–943. - PubMed
-
- Wyman C, et al. DNA double-strand break repair: all's well that ends well. Annu. Rev. Genet. 2006;40:363–383. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials

