Model calcium sensors for network homeostasis: sensor and readout parameter analysis from a database of model neuronal networks
- PMID: 20130178
- PMCID: PMC2851246
- DOI: 10.1523/JNEUROSCI.3098-09.2010
Model calcium sensors for network homeostasis: sensor and readout parameter analysis from a database of model neuronal networks
Abstract
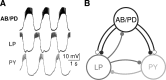
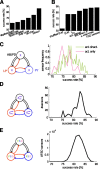
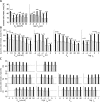
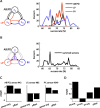
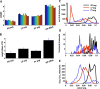
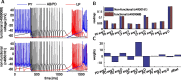
In activity-dependent homeostatic regulation (ADHR) of neuronal and network properties, the intracellular Ca(2+) concentration is a good candidate for sensing activity levels because it is correlated with the electrical activity of the cell. Previous ADHR models, developed with abstract activity sensors for model pyloric neurons and networks of the crustacean stomatogastric ganglion, showed that functional activity can be maintained by a regulation mechanism that senses activity levels solely from Ca(2+). At the same time, several intracellular pathways have been discovered for Ca(2+)-dependent regulation of ion channels. To generate testable predictions for dynamics of these signaling pathways, we undertook a parameter study of model Ca(2+) sensors across thousands of model pyloric networks. We found that an optimal regulation signal can be generated for 86% of model networks with a sensing mechanism that activates with a time constant of 1 ms and that inactivates within 1 s. The sensor performed robustly around this optimal point and did not need to be specific to the role of the cell. When multiple sensors with different time constants were used, coverage extended to 88% of the networks. Without changing the sensors, it extended to 95% of the networks by letting the sensors affect the readout nonlinearly. Specific to this pyloric network model, the sensor of the follower pyloric constrictor cell was more informative than the pacemaker anterior burster cell for producing a regulatory signal. Conversely, a global signal indicating network activity that was generated by summing the sensors in individual cells was less informative for regulation.
Figures

Similar articles
-
Calcium Sensors in Neuronal Function and Dysfunction.Cold Spring Harb Perspect Biol. 2019 May 1;11(5):a035154. doi: 10.1101/cshperspect.a035154. Cold Spring Harb Perspect Biol. 2019. PMID: 30833454 Free PMC article. Review.
-
Neuromodulatory changes in short-term synaptic dynamics may be mediated by two distinct mechanisms of presynaptic calcium entry.J Comput Neurosci. 2012 Dec;33(3):573-85. doi: 10.1007/s10827-012-0402-z. Epub 2012 Jun 19. J Comput Neurosci. 2012. PMID: 22710936 Free PMC article.
-
Co-variation of ionic conductances supports phase maintenance in stomatogastric neurons.J Comput Neurosci. 2012 Aug;33(1):77-95. doi: 10.1007/s10827-011-0375-3. Epub 2011 Dec 3. J Comput Neurosci. 2012. PMID: 22134522 Free PMC article.
-
Membrane resonance in bursting pacemaker neurons of an oscillatory network is correlated with network frequency.J Neurosci. 2009 May 20;29(20):6427-35. doi: 10.1523/JNEUROSCI.0545-09.2009. J Neurosci. 2009. PMID: 19458214 Free PMC article.
-
Regulation of transmitter release by Ca(2+) and synaptotagmin: insights from a large CNS synapse.Trends Neurosci. 2011 May;34(5):237-46. doi: 10.1016/j.tins.2011.02.006. Epub 2011 Mar 24. Trends Neurosci. 2011. PMID: 21439657 Review.
Cited by
-
Compensation for variable intrinsic neuronal excitability by circuit-synaptic interactions.J Neurosci. 2010 Jul 7;30(27):9145-56. doi: 10.1523/JNEUROSCI.0980-10.2010. J Neurosci. 2010. PMID: 20610748 Free PMC article.
-
Activation of high and low affinity dopamine receptors generates a closed loop that maintains a conductance ratio and its activity correlate.Front Neural Circuits. 2013 Oct 22;7:169. doi: 10.3389/fncir.2013.00169. eCollection 2013. Front Neural Circuits. 2013. PMID: 24155696 Free PMC article.
-
Pumilio-2 regulates translation of Nav1.6 to mediate homeostasis of membrane excitability.J Neurosci. 2013 Jun 5;33(23):9644-54. doi: 10.1523/JNEUROSCI.0921-13.2013. J Neurosci. 2013. PMID: 23739961 Free PMC article.
-
Myocyte enhancer factor-2 and p300 interact to regulate the expression of homeostatic regulator Pumilio in Drosophila.Eur J Neurosci. 2019 Jul;50(1):1727-1740. doi: 10.1111/ejn.14357. Epub 2019 Feb 21. Eur J Neurosci. 2019. PMID: 30687963 Free PMC article.
-
Activity-dependent compensation of cell size is vulnerable to targeted deletion of ion channels.Sci Rep. 2020 Sep 29;10(1):15989. doi: 10.1038/s41598-020-72977-6. Sci Rep. 2020. PMID: 32994529 Free PMC article.
References
-
- Abbott L, LeMasson G. Analysis of neuron models with dynamically regulated conductances. Neural Comp. 1993;5:823–842.
-
- An WF, Bowlby MR, Betty M, Cao J, Ling HP, Mendoza G, Hinson JW, Mattsson KI, Strassle BW, Trimmer JS, Rhodes KJ. Modulation of A-type potassium channels by a family of calcium sensors. Nature. 2000;403:553–556. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous
