Structure of epi-isozizaene synthase from Streptomyces coelicolor A3(2), a platform for new terpenoid cyclization templates
- PMID: 20131801
- PMCID: PMC2840623
- DOI: 10.1021/bi902088z
Structure of epi-isozizaene synthase from Streptomyces coelicolor A3(2), a platform for new terpenoid cyclization templates
Abstract
The X-ray crystal structure of recombinant epi-isozizaene synthase (EIZS), a sesquiterpene cyclase from Streptomyces coelicolor A3(2), has been determined at 1.60 A resolution. Specifically, the structure of wild-type EIZS is that of its closed conformation in complex with three Mg(2+) ions, inorganic pyrophosphate (PP(i)), and the benzyltriethylammonium cation (BTAC). Additionally, the structure of D99N EIZS has been determined in an open, ligand-free conformation at 1.90 A resolution. Comparison of these two structures provides the first view of conformational changes required for substrate binding and catalysis in a bacterial terpenoid cyclase. Moreover, the binding interactions of BTAC may mimic those of a carbocation intermediate in catalysis. Accordingly, the aromatic rings of F95, F96, and F198 appear to be well-oriented to stabilize carbocation intermediates in the cyclization cascade through cation-pi interactions. Mutagenesis of aromatic residues in the enzyme active site results in the production of alternative sesquiterpene product arrays due to altered modes of stabilization of carbocation intermediates as well as altered templates for the cyclization of farnesyl diphosphate. Accordingly, the 1.64 A resolution crystal structure of F198A EIZS in a complex with three Mg(2+) ions, PP(i), and BTAC reveals an alternative binding orientation of BTAC; alternative binding orientations of a carbocation intermediate could lead to the formation of alternative products. Finally, the crystal structure of wild-type EIZS in a complex with four Hg(2+) ions has been determined at 1.90 A resolution, showing that metal binding triggers a significant conformational change of helix G to cap the active site.
Figures




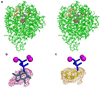

References
-
- Lesburg CA, Caruthers JM, Paschall CM, Christianson DW. Managing and manipulating carbocations in biology: terpenoid cyclase structure and mechanism. Curr. Opin. Struct. Biol. 1998;8:695–703. - PubMed
-
- Christianson DW. Structural biology and chemistry of the terpenoid cyclases. Chem. Rev. 2006;106:3412–3442. - PubMed
-
- Tholl D. Terpene synthases and the regulation, diversity and biological roles of terpene metabolism. Curr. Opin. Plant Biol. 2006;9:297–304. - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
- Actions
- Actions
- Actions
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

