A 13-Oxo-9,10-epoxytridecenoate phospholipid analogue of the genotoxic 4,5-epoxy-2E-decenal: detection in vivo, chemical synthesis, and adduction with DNA
- PMID: 20131875
- PMCID: PMC2848942
- DOI: 10.1021/tx9002484
A 13-Oxo-9,10-epoxytridecenoate phospholipid analogue of the genotoxic 4,5-epoxy-2E-decenal: detection in vivo, chemical synthesis, and adduction with DNA
Abstract
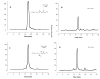
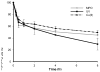
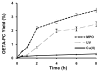
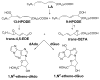
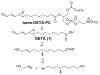
Often guided by analogy with nonphospholipid products from oxidative cleavage of polyunsaturated fatty acids, we previously identified a variety of biologically active oxidatively truncated phospholipids. Previously, 4,5-epoxy-2(E)-decenal (4,5-EDE) was found to be produced by oxidative cleavage of 13-(S)-hydroperoxy-9,11-(Z,E)-octadeca-dienoic acid (13-HPODE). 4,5-EDE reacts with deoxy-adenosine (dAdo) and deoxy-guanosine (dGuo) to form mutagenic etheno derivatives. We hypothesized that a functionally similar and potentially mutagenic compound, that is, 13-oxo-9,10-epoxytridecenoic acid (OETA), would be generated from 9-HPODE through an analogous fragmentation. We expected that an ester of 2-lysophosphatidylcoline (PC), OETA-PC, would be produced by oxidative cleavage of 9-HPODE-PC in biological membranes. An efficient, unambiguous total synthesis of trans-OETA-PC was first executed to provide a standard that could facilitate the identification of this phospholipid epoxyalkenal that was shown to be produced during oxidation of the linoleic acid ester of 2-lysoPC. Finally, trans-OETA-PC was detected in a lipid extract from rat retina. The identity of the naturally occurring oxidatively truncated phospholipid was further confirmed by derivatization with methoxylamine that produced characteristic mono and bis adducts. The average amount of trans-OETA-PC in rat retina, 0.33 pmol, is relatively low as compared to other oxidatively truncated PCs, for example, the 4-hydroxy-7-oxohept-5-enoic acid PC ester (2.5 pmol) or the 4-keto-7-oxohept-5-enoic acid PC ester (1.7 pmol), derived from the docosahexaenoic acid ester of 2-lysoPC. This, most likely, is because docosahexaenoate PCs are particularly abundant in the retina as compared to the linoleate PC ester precursor of OETA-PC. As predicted by analogy with 4,5-EDE, OETA-PC reacts with dAdo and dGuo, as well as with DNA, to form mutagenic etheno adducts.
Figures
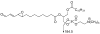
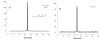





Similar articles
-
Oxidative fragmentation of hydroxy octadecadienoates generates biologically active gamma-hydroxyalkenals.J Am Chem Soc. 2004 May 12;126(18):5699-708. doi: 10.1021/ja038756w. J Am Chem Soc. 2004. PMID: 15125662
-
Oxidatively truncated docosahexaenoate phospholipids: total synthesis, generation, and Peptide adduction chemistry.J Org Chem. 2003 May 16;68(10):3749-61. doi: 10.1021/jo026721t. J Org Chem. 2003. PMID: 12737551
-
Liquid chromatography/mass spectrometry analysis of bifunctional electrophiles and DNA adducts from vitamin C mediated decomposition of 15-hydroperoxyeicosatetraenoic acid.Rapid Commun Mass Spectrom. 2005;19(6):849-58. doi: 10.1002/rcm.1854. Rapid Commun Mass Spectrom. 2005. PMID: 15723435
-
Oxidative DNA damage and cardiovascular disease.Trends Cardiovasc Med. 2001 Apr-May;11(3-4):148-55. doi: 10.1016/s1050-1738(01)00094-9. Trends Cardiovasc Med. 2001. PMID: 11686005 Review.
-
Lipid hydroperoxide-mediated DNA damage.Exp Gerontol. 2001 Sep;36(9):1473-81. doi: 10.1016/s0531-5565(01)00133-4. Exp Gerontol. 2001. PMID: 11525870 Review.
Cited by
-
Simplified synthesis of oxidized phospholipids on alkyl-amide scaffold.MethodsX. 2025 Mar 27;14:103288. doi: 10.1016/j.mex.2025.103288. eCollection 2025 Jun. MethodsX. 2025. PMID: 40230553 Free PMC article.
-
Synthesis of the four stereoisomers of 2,3-epoxy-4-hydroxynonanal and their reactivity with deoxyguanosine.Org Biomol Chem. 2011 Mar 21;9(6):1960-71. doi: 10.1039/c0ob00546k. Epub 2011 Jan 24. Org Biomol Chem. 2011. PMID: 21258716 Free PMC article.
References
-
- Greenberg ME, Li XM, Gugiu BG, Gu X, Qin J, Salomon RG, Hazen SL. The lipid whisker model of the structure of oxidized cell membranes. The Journal of biological chemistry. 2008;283:2385–2396. - PubMed
-
- Podrez EA, Poliakov E, Shen Z, Zhang R, Deng Y, Sun M, Finton PJ, Shan L, Gugiu B, Fox PL, Hoff HF, Salomon RG, Hazen SL. Identification of a novel family of oxidized phospholipids that serve as ligands for the macrophage scavenger receptor CD36. J Biol Chem. 2002;277:38503–38516. - PubMed
-
- Sun M, Finnemann SC, Febbraio M, Shan L, Annangudi SP, Podrez EA, Hoppe G, Darrow R, Organisciak DT, Salomon RG, Silverstein RL, Hazen SL. Light-induced oxidation of photoreceptor outer segment phospholipids generates ligands for CD36-mediated phagocytosis by retinal pigment epithelium: a potential mechanism for modulating outer segment phagocytosis under oxidant stress conditions. J Biol Chem. 2006;281:4222–4230. - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

