Engineered vascularized bone grafts
- PMID: 20133604
- PMCID: PMC2840421
- DOI: 10.1073/pnas.0905445107
Engineered vascularized bone grafts
Erratum in
-
Correction for Tsigkou et al., Engineered vascularized bone grafts.Proc Natl Acad Sci U S A. 2018 Sep 11;115(37):E8811. doi: 10.1073/pnas.1813106115. Epub 2018 Sep 4. Proc Natl Acad Sci U S A. 2018. PMID: 30181266 Free PMC article. No abstract available.
Abstract
Clinical protocols utilize bone marrow to seed synthetic and decellularized allogeneic bone grafts for enhancement of scaffold remodeling and fusion. Marrow-derived cytokines induce host neovascularization at the graft surface, but hypoxic conditions cause cell death at the core. Addition of cellular components that generate an extensive primitive plexus-like vascular network that would perfuse the entire scaffold upon anastomosis could potentially yield significantly higher-quality grafts. We used a mouse model to develop a two-stage protocol for generating vascularized bone grafts using mesenchymal stem cells (hMSCs) from human bone marrow and umbilical cord-derived endothelial cells. The endothelial cells formed tube-like structures and subsequently networks throughout the bone scaffold 4-7 days after implantation. hMSCs were essential for stable vasculature both in vitro and in vivo; however, contrary to expectations, vasculature derived from hMSCs briefly cultured in medium designed to maintain a proliferative, nondifferentiated state was more extensive and stable than that with hMSCs with a TGF-beta-induced smooth muscle cell phenotype. Anastomosis occurred by day 11, with most hMSCs associating closely with the network. Although initially immature and highly permeable, at 4 weeks the network was mature. Initiation of scaffold mineralization had also occurred by this period. Some human-derived vessels were still present at 5 months, but the majority of the graft vasculature had been functionally remodeled with host cells. In conclusion, clinically relevant progenitor sources for pericytes and endothelial cells can serve to generate highly functional microvascular networks for tissue engineered bone grafts.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


 ;
;  ,
,  . ∗P < 0.05.)
. ∗P < 0.05.)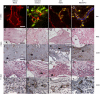
 ;
;  .)
.)
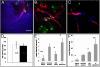
 .)
.)
 ;
;  .)
.)References
-
- Muschler GF, Nakamoto C, Griffith LG. Engineering principles of clinical cell-based tissue engineering. J Bone Joint Surg Am. 2004;86-A(7):1541–1558. - PubMed
-
- Folkman J, Haudenschild C. Angiogenesis in vitro. Nature. 1980;288(5791):551–556. - PubMed
-
- Sato N, et al. Development of capillary networks from rat microvascular fragments in vitro: The role of myofibroblastic cells. Microvasc Res. 1987;33(2):194–210. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources

