DJ-1 protects the nigrostriatal axis from the neurotoxin MPTP by modulation of the AKT pathway
- PMID: 20133695
- PMCID: PMC2840364
- DOI: 10.1073/pnas.0914876107
DJ-1 protects the nigrostriatal axis from the neurotoxin MPTP by modulation of the AKT pathway
Abstract
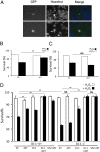
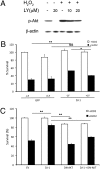
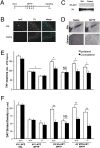
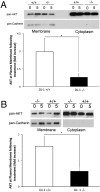
Loss-of-function DJ-1 (PARK7) mutations have been linked with a familial form of early onset Parkinson disease. Numerous studies have supported the role of DJ-1 in neuronal survival and function. Our initial studies using DJ-1-deficient neurons indicated that DJ-1 specifically protects the neurons against the damage induced by oxidative injury in multiple neuronal types and degenerative experimental paradigms, both in vitro and in vivo. However, the manner by which oxidative stress-induced death is ameliorated by DJ-1 is not completely clear. We now present data that show the involvement of DJ-1 in modulation of AKT, a major neuronal prosurvival pathway induced upon oxidative stress. We provide evidence that DJ-1 promotes AKT phosphorylation in response to oxidative stress induced by H(2)O(2) in vitro and in vivo following 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP) treatment. Moreover, we show that DJ-1 is necessary for normal AKT-mediated protective effects, which can be bypassed by expression of a constitutively active form of AKT. Taken together, these data suggest that DJ-1 is crucial for full activation of AKT upon oxidative injury, which serves as one explanation for the protective effects of DJ-1.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

References
-
- Bonifati V, et al. Mutations in the DJ-1 gene associated with autosomal recessive early-onset parkinsonism. Science. 2003;299:256–259. - PubMed
-
- Abou-Sleiman PM, Healy DG, Quinn N, Lees AJ, Wood NW. The role of pathogenic DJ-1 mutations in Parkinson’s disease. Ann Neurol. 2003;54:283–286. - PubMed
-
- Gu L, et al. Involvement of ERK1/2 signaling pathway in DJ-1-induced neuroprotection against oxidative stress. Biochem Biophys Res Commun. 2009;383:469–474. - PubMed
-
- Inden M, et al. PARK7 DJ-1 protects against degeneration of nigral dopaminergic neurons in Parkinson’s disease rat model. Neurobiol Dis. 2006;24:144–158. - PubMed
-
- Lev N, et al. DJ-1 protects against dopamine toxicity. J Neural Transm. 2009;116:151–160. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

