Asymmetric receptor contact is required for tyrosine autophosphorylation of fibroblast growth factor receptor in living cells
- PMID: 20133753
- PMCID: PMC2840318
- DOI: 10.1073/pnas.0914157107
Asymmetric receptor contact is required for tyrosine autophosphorylation of fibroblast growth factor receptor in living cells
Abstract
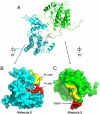
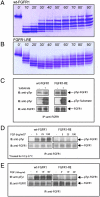
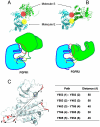
Tyrosine autophosphorylation of receptor tyrosine kinases plays a critical role in regulation of kinase activity and in recruitment and activation of intracellular signaling pathways. Autophosphorylation is mediated by a sequential and precisely ordered intermolecular (trans) reaction. In this report we present structural and biochemical experiments demonstrating that formation of an asymmetric dimer between activated FGFR1 kinase domains is required for transphosphorylation of FGFR1 in FGF-stimulated cells. Transphosphorylation is mediated by specific asymmetric contacts between the N-lobe of one kinase molecule, which serves as an active enzyme, and specific docking sites on the C-lobe of a second kinase molecule, which serves a substrate. Pathological loss-of-function mutations or oncogenic activating mutations in this interface may hinder or facilitate asymmetric dimer formation and transphosphorylation, respectively. The experiments presented in this report provide the molecular basis underlying the control of transphosphorylation of FGF receptors and other receptor tyrosine kinases.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

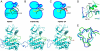
References
-
- Schlessinger J. Signal transduction by allosteric receptor oligomerization. Trends Biochem Sci. 1988;13:443–447. - PubMed
-
- Schlessinger J. Cell signaling by receptor tyrosine kinases. Cell. 2000;10:211–225. - PubMed
-
- Schlessinger J, Lemmon MA. SH2 and PTB domains in tyrosine kinase signaling. Sci STKE. 2003;19:RE12. - PubMed
-
- Schlessinger J, Ullrich A. Growth factor signaling by receptor tyrosine kinases. Neuron. 1992;9:383–391. - PubMed
-
- Lemmon MA, Schlessinger J. Regulation of signal transduction and signal diveristy by receptor oligomerization. Trends Biochem Sci. 1994;19:459–463. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

