Evidence for a functional role of IgE anticitrullinated protein antibodies in rheumatoid arthritis
- PMID: 20133791
- PMCID: PMC2823893
- DOI: 10.1073/pnas.0913054107
Evidence for a functional role of IgE anticitrullinated protein antibodies in rheumatoid arthritis
Retraction in
-
Retraction for Schuerwegh et al., Evidence for a functional role of IgE anticitrullinated protein antibodies in rheumatoid arthritis.Proc Natl Acad Sci U S A. 2013 Dec 10;110(50):20345. doi: 10.1073/pnas.1320459110. Epub 2013 Nov 25. Proc Natl Acad Sci U S A. 2013. PMID: 24277813 Free PMC article. No abstract available.
Abstract
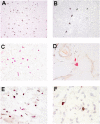
Rheumatoid arthritis (RA) is a systemic autoimmune disease involving inflammation of the joints. Among the autoantibodies described in RA, anticitrullinated protein antibodies (ACPAs) are highly specific and predictive for RA. In addition, ACPAs have been implicated in the pathogenesis of RA. However, a direct functional response of immune cells from ACPA(+) RA patients toward citrullinated proteins has not been demonstrated. In this study, we show that exposure to citrullinated antigens leads to activation of basophils from ACPA(+) RA patients within 20 minutes. This was not observed after exposure of basophils to noncitrullinated control antigens or after stimulation of basophils from ACPA(-) RA patients and healthy controls. Basophil activation was correlated with the binding of citrullinated proteins to basophils. Furthermore, serum from ACPA(+) RA patients in contrast to that from ACPA(-) RA patients could specifically sensitize human FcepsilonRI expressing rat basophil cells (RBL), enabling activation by citrullinated proteins. Mast cell degranulation products such as histamine levels were enhanced in synovial fluid of ACPA(+) RA patients as compared with ACPA(-) RA and osteoarthritis patients. In addition, histamine levels in synovial fluid from ACPA(+) RA patients correlated with IgE levels, suggesting degranulation of mast cells by cross-linking IgE. Immunohistochemistry on synovial biopsies demonstrated an increased number of degranulated CD117(+) mast cells in ACPA(+) RA patients; IgE and FcepsilonRI expression in synovial mast cells from ACPA(+) RA patients was increased. In conclusion, our results show an immunological response of immune cells from ACPA(+) RA patients in a citrulline-specific manner. Moreover, these data indicate a role for IgE-ACPAs and FcepsilonRI-positive cells in the pathogenesis of RA.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

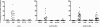
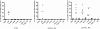

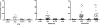


References
-
- Schellekens GA, et al. The diagnostic properties of rheumatoid arthritis antibodies recognizing a cyclic citrullinated peptide. Arthritis Rheum. 2000;43:155–163. - PubMed
-
- van Gaalen FA, et al. Autoantibodies to cyclic citrullinated peptides predict progression to rheumatoid arthritis in patients with undifferentiated arthritis: A prospective cohort study. Arthritis Rheum. 2004;50:709–715. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

