Influence of lever structure on myosin 5a walking
- PMID: 20133809
- PMCID: PMC2823865
- DOI: 10.1073/pnas.0906907107
Influence of lever structure on myosin 5a walking
Abstract
Using electron microscopy and image processing, we have observed myosin 5a modified with lever arms of different lengths (four, six, and eight calmodulin-binding IQ domains) and orientations walking along actin filaments. Step lengths were dependent on lever length: 8IQ > 6IQ > 4IQ, which is consistent with myosin 5a having evolved to walk straight along actin. Lead heads were mostly in the prepowerstroke state, tethered there by the trail head. However, improved image processing showed that in 5-10% of molecules the lead motor was in the postpowerstroke state. This is a unique attached state of myosin, where the motor domain has completed its powerstroke at the expense of severe lever distortion, but with little cargo movement. Postpowerstroke lead heads were seen in both wild-type and modified lever molecules, mostly where there was least strain. These data allow the strain dependence of the equilibrium between pre- and postpowerstroke conformations to be measured. Slow rates of ADP dissociation observed from lead heads of these molecules can be explained by the unfavorable equilibrium between the pre- and postpowerstroke conformations preceding ADP loss.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
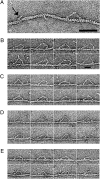
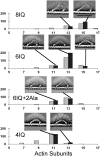

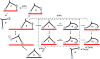
 . T, ATP; D, ADP; P, phosphate.
. T, ATP; D, ADP; P, phosphate.Similar articles
-
The SAH domain extends the functional length of the myosin lever.Proc Natl Acad Sci U S A. 2009 Dec 29;106(52):22193-8. doi: 10.1073/pnas.0909851106. Epub 2009 Dec 15. Proc Natl Acad Sci U S A. 2009. PMID: 20018767 Free PMC article.
-
Neck length and processivity of myosin V.J Biol Chem. 2003 Aug 1;278(31):29201-7. doi: 10.1074/jbc.M303662200. Epub 2003 May 11. J Biol Chem. 2003. PMID: 12740393
-
Switch 1 mutation S217A converts myosin V into a low duty ratio motor.J Biol Chem. 2009 Jan 23;284(4):2138-49. doi: 10.1074/jbc.M805530200. Epub 2008 Nov 12. J Biol Chem. 2009. PMID: 19008235 Free PMC article.
-
How Myosin Generates Force on Actin Filaments.Trends Biochem Sci. 2016 Dec;41(12):989-997. doi: 10.1016/j.tibs.2016.09.006. Epub 2016 Oct 4. Trends Biochem Sci. 2016. PMID: 27717739 Free PMC article. Review.
-
Cooperativity of myosin molecules through strain-dependent chemistry.Philos Trans R Soc Lond B Biol Sci. 2000 Apr 29;355(1396):529-38. doi: 10.1098/rstb.2000.0594. Philos Trans R Soc Lond B Biol Sci. 2000. PMID: 10836506 Free PMC article. Review.
Cited by
-
Functions of class V myosins in neurons.J Biol Chem. 2013 Oct 4;288(40):28428-34. doi: 10.1074/jbc.R113.514497. Epub 2013 Aug 29. J Biol Chem. 2013. PMID: 23990471 Free PMC article. Review.
-
The path to visualization of walking myosin V by high-speed atomic force microscopy.Biophys Rev. 2014;6(3-4):237-260. doi: 10.1007/s12551-014-0141-7. Epub 2014 Jun 18. Biophys Rev. 2014. PMID: 25505494 Free PMC article.
-
Sequence Alignment-Based Prediction of Myosin 7A: Structural Implications and Protein Interactions.Int J Mol Sci. 2024 Mar 16;25(6):3365. doi: 10.3390/ijms25063365. Int J Mol Sci. 2024. PMID: 38542339 Free PMC article.
-
Two single-headed myosin V motors bound to a tetrameric adapter protein form a processive complex.J Cell Biol. 2011 Nov 14;195(4):631-41. doi: 10.1083/jcb.201106146. J Cell Biol. 2011. PMID: 22084309 Free PMC article.
-
Switching of myosin-V motion between the lever-arm swing and brownian search-and-catch.Nat Commun. 2012 Jul 17;3:956. doi: 10.1038/ncomms1934. Nat Commun. 2012. PMID: 22805563
References
-
- Cheney RE, et al. Brain myosin-V is a two-headed unconventional myosin with motor activity. Cell. 1993;75:13–23. - PubMed
-
- Yildiz A, et al. Myosin V walks hand-over-hand: Single fluorophore imaging with 1.5-nm localization. Science. 2003;300:2061–2065. - PubMed
-
- Forkey JN, Quinlan ME, Shaw MA, Corrie JET, Goldman YE. Three-dimensional structural dynamics of myosin V by single-molecule fluorescence polarization. Nature. 2003;422:399–404. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

