Effects of CXCR4 gene transfer on cardiac function after ischemia-reperfusion injury
- PMID: 20133817
- PMCID: PMC2843462
- DOI: 10.2353/ajpath.2010.090451
Effects of CXCR4 gene transfer on cardiac function after ischemia-reperfusion injury
Abstract
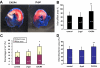
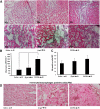
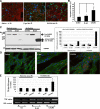
Acute coronary occlusion is the leading cause of death in the Western world. There is an unmet need for the development of treatments to limit the extent of myocardial infarction (MI) during the acute phase of occlusion. Recently, investigators have focused on the use of a chemokine, CXCL12, the only identified ligand for CXCR4, as a new therapeutic modality to recruit stem cells to individuals suffering from MI. Here, we examined the effects of overexpression of CXCR4 by gene transfer on MI. Adenoviruses carrying the CXCR4 gene were injected into the rat heart one week before ligation of the left anterior descending coronary artery followed by 24 hours reperfusion. Cardiac function was assessed by echocardiography couple with 2,3,5-Triphenyltetrazolium chloride staining to measure MI size. In comparison with control groups, rats receiving Ad-CXCR4 displayed an increase in infarct area (13.5% +/- 4.1%) and decreased fractional shortening (38% +/- 5%). Histological analysis revealed a significant increase in CXCL12 and tumor necrosis factor-alpha expression in ischemic area of CXCR4 overexpressed hearts. CXCR4 overexpression was associated with increased influx of inflammatory cells and enhanced cardiomyocyte apoptosis in the infarcted heart. These data suggest that in our model overexpressing CXCR4 appears to enhance ischemia/reperfusion injury possibly due to enhanced recruitment of inflammatory cells, increased tumor necrosis factor-alpha production, and activation of cell death/apoptotic pathways.
Figures



Similar articles
-
Stromal cell-derived factor-1α signals via the endothelium to protect the heart against ischaemia-reperfusion injury.J Mol Cell Cardiol. 2019 Mar;128:187-197. doi: 10.1016/j.yjmcc.2019.02.002. Epub 2019 Feb 7. J Mol Cell Cardiol. 2019. PMID: 30738798 Free PMC article.
-
Genetically manipulated progenitor cell sheet with diprotin A improves myocardial function and repair of infarcted hearts.Am J Physiol Heart Circ Physiol. 2010 Nov;299(5):H1339-47. doi: 10.1152/ajpheart.00592.2010. Epub 2010 Aug 27. Am J Physiol Heart Circ Physiol. 2010. PMID: 20802132 Free PMC article.
-
Bone-derived Nestin-positive mesenchymal stem cells improve cardiac function via recruiting cardiac endothelial cells after myocardial infarction.Stem Cell Res Ther. 2019 Apr 27;10(1):127. doi: 10.1186/s13287-019-1217-x. Stem Cell Res Ther. 2019. PMID: 31029167 Free PMC article.
-
Double-edged role of the CXCL12/CXCR4 axis in experimental myocardial infarction.J Am Coll Cardiol. 2011 Nov 29;58(23):2415-23. doi: 10.1016/j.jacc.2011.08.033. J Am Coll Cardiol. 2011. PMID: 22115649
-
Danshensu accelerates angiogenesis after myocardial infarction in rats and promotes the functions of endothelial progenitor cells through SDF-1α/CXCR4 axis.Eur J Pharmacol. 2017 Nov 5;814:274-282. doi: 10.1016/j.ejphar.2017.08.035. Epub 2017 Aug 31. Eur J Pharmacol. 2017. PMID: 28864209
Cited by
-
The CXCR4/CXCR7/SDF-1 pathway contributes to the pathogenesis of Shiga toxin-associated hemolytic uremic syndrome in humans and mice.J Clin Invest. 2012 Feb;122(2):759-76. doi: 10.1172/JCI57313. Epub 2012 Jan 9. J Clin Invest. 2012. PMID: 22232208 Free PMC article.
-
Expression of chemokine receptor-4 in bone marrow mesenchymal stem cells on experimental rat abdominal aortic aneurysms and the migration of bone marrow mesenchymal stem cells with stromal-derived factor-1.Kaohsiung J Med Sci. 2014 May;30(5):224-8. doi: 10.1016/j.kjms.2013.12.005. Epub 2014 Feb 16. Kaohsiung J Med Sci. 2014. PMID: 24751384 Free PMC article.
-
The CXCL12/CXCR4 chemokine ligand/receptor axis in cardiovascular disease.Front Physiol. 2014 Jun 11;5:212. doi: 10.3389/fphys.2014.00212. eCollection 2014. Front Physiol. 2014. PMID: 24966838 Free PMC article. Review.
-
Stromal cell-derived factor-1α signals via the endothelium to protect the heart against ischaemia-reperfusion injury.J Mol Cell Cardiol. 2019 Mar;128:187-197. doi: 10.1016/j.yjmcc.2019.02.002. Epub 2019 Feb 7. J Mol Cell Cardiol. 2019. PMID: 30738798 Free PMC article.
-
Cardiac gene therapy: are we there yet?Gene Ther. 2016 Aug;23(8-9):635-48. doi: 10.1038/gt.2016.43. Epub 2016 Apr 29. Gene Ther. 2016. PMID: 27128687 Review.
References
-
- Tissier R, Berdeaux A, Ghaleh B, Couvreur N, Krieg T, Cohen MV, Downey JM. Making the heart resistant to infarction: how can we further decrease infarct size? Front Biosci. 2008;13:284–301. - PubMed
-
- Carden DL, Granger DN. Pathophysiology of ischaemia-reperfusion injury. J Pathol. 2000;190:255–266. - PubMed
-
- Vermeiren GL, Claeys MJ, Van Bockstaele D, Grobben B, Slegers H, Bossaert L, Jorens PG. Reperfusion injury after focal myocardial ischaemia: polymorphonuclear leukocyte activation and its clinical implications. Resuscitation. 2000;45:35–61. - PubMed
-
- Gao L, Yin H, Smith S RJ, Chao L, Chao J. Role of kallistatin in prevention of cardiac remodeling after chronic myocardial infarction. Lab Invest. 2008;88:1157–1166. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

