Nanowire transistor arrays for mapping neural circuits in acute brain slices
- PMID: 20133836
- PMCID: PMC2808222
- DOI: 10.1073/pnas.0914737107
Nanowire transistor arrays for mapping neural circuits in acute brain slices
Abstract
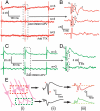
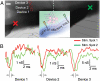
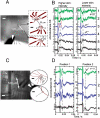
Revealing the functional connectivity in natural neuronal networks is central to understanding circuits in the brain. Here, we show that silicon nanowire field-effect transistor (Si NWFET) arrays fabricated on transparent substrates can be reliably interfaced to acute brain slices. NWFET arrays were readily designed to record across a wide range of length scales, while the transparent device chips enabled imaging of individual cell bodies and identification of areas of healthy neurons at both upper and lower tissue surfaces. Simultaneous NWFET and patch clamp studies enabled unambiguous identification of action potential signals, with additional features detected at earlier times by the nanodevices. NWFET recording at different positions in the absence and presence of synaptic and ion-channel blockers enabled assignment of these features to presynaptic firing and postsynaptic depolarization from regions either close to somata or abundant in dendritic projections. In all cases, the NWFET signal amplitudes were from 0.3-3 mV. In contrast to conventional multielectrode array measurements, the small active surface of the NWFET devices, approximately 0.06 microm(2), provides highly localized multiplexed measurements of neuronal activities with demonstrated sub-millisecond temporal resolution and, significantly, better than 30 microm spatial resolution. In addition, multiplexed mapping with 2D NWFET arrays revealed spatially heterogeneous functional connectivity in the olfactory cortex with a resolution surpassing substantially previous electrical recording techniques. Our demonstration of simultaneous high temporal and spatial resolution recording, as well as mapping of functional connectivity, suggest that NWFETs can become a powerful platform for studying neural circuits in the brain.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


Comment in
-
Nanowire platform for mapping neural circuits.Proc Natl Acad Sci U S A. 2010 Mar 9;107(10):4489-90. doi: 10.1073/pnas.1000450107. Epub 2010 Mar 1. Proc Natl Acad Sci U S A. 2010. PMID: 20194753 Free PMC article. No abstract available.
Similar articles
-
Nanowire platform for mapping neural circuits.Proc Natl Acad Sci U S A. 2010 Mar 9;107(10):4489-90. doi: 10.1073/pnas.1000450107. Epub 2010 Mar 1. Proc Natl Acad Sci U S A. 2010. PMID: 20194753 Free PMC article. No abstract available.
-
Flexible electrical recording from cells using nanowire transistor arrays.Proc Natl Acad Sci U S A. 2009 May 5;106(18):7309-13. doi: 10.1073/pnas.0902752106. Epub 2009 Apr 13. Proc Natl Acad Sci U S A. 2009. PMID: 19365078 Free PMC article.
-
Electrical recording from hearts with flexible nanowire device arrays.Nano Lett. 2009 Feb;9(2):914-8. doi: 10.1021/nl900096z. Nano Lett. 2009. PMID: 19170614 Free PMC article.
-
Construction of functional neuronal circuitry in the olfactory bulb.Semin Cell Dev Biol. 2014 Nov;35:180-8. doi: 10.1016/j.semcdb.2014.07.012. Epub 2014 Jul 30. Semin Cell Dev Biol. 2014. PMID: 25084319 Review.
-
Mapping functional connectivity in barrel-related columns reveals layer- and cell type-specific microcircuits.Brain Struct Funct. 2007 Sep;212(2):107-19. doi: 10.1007/s00429-007-0147-z. Epub 2007 Jun 26. Brain Struct Funct. 2007. PMID: 17717691 Review.
Cited by
-
Comparative Characterization of NWFET and FinFET Transistor Structures Using TCAD Modeling.Micromachines (Basel). 2022 Aug 11;13(8):1293. doi: 10.3390/mi13081293. Micromachines (Basel). 2022. PMID: 36014213 Free PMC article.
-
Mesh Nanoelectronics: Seamless Integration of Electronics with Tissues.Acc Chem Res. 2018 Feb 20;51(2):309-318. doi: 10.1021/acs.accounts.7b00547. Epub 2018 Jan 30. Acc Chem Res. 2018. PMID: 29381054 Free PMC article.
-
Interfacing with the Brain: How Nanotechnology Can Contribute.ACS Nano. 2025 Mar 25;19(11):10630-10717. doi: 10.1021/acsnano.4c10525. Epub 2025 Mar 10. ACS Nano. 2025. PMID: 40063703 Free PMC article. Review.
-
High spatial and temporal resolution wide-field imaging of neuron activity using quantum NV-diamond.Sci Rep. 2012;2:401. doi: 10.1038/srep00401. Epub 2012 May 9. Sci Rep. 2012. PMID: 22574249 Free PMC article.
-
Nanowire platform for mapping neural circuits.Proc Natl Acad Sci U S A. 2010 Mar 9;107(10):4489-90. doi: 10.1073/pnas.1000450107. Epub 2010 Mar 1. Proc Natl Acad Sci U S A. 2010. PMID: 20194753 Free PMC article. No abstract available.
References
-
- Shepherd GM. The Synaptic Organization of the Brain. 5th Ed. New York: Oxford Univ Press, Inc.; 2004. Ch. 1, 5, 10, 11.
-
- Taketani M, Baudry M. Advances in Network Electrophysiology Using Multi-Electrode Arrays. New York: Springer Science+Business Media, Inc.; 2006.
-
- Hutzler M, et al. High-resolution multitransistor array recording of electrical field potentials in cultured brain slices. J Neurophysiol. 2006;96:1638–1645. - PubMed
-
- Egert U, Heck D, Aertsen A. Two-dimensional monitoring of spiking networks in acute brain slices. Exp Brain Res. 2002;142:268–274. - PubMed
-
- Wirth C, Lüscher HR. Spatiotemporal evolution of excitation and inhibition in the rat barrel cortex investigated with multielectrode arrays. J Neurophysiol. 2004;91:1635–1647. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

