Rational design of dualsteric GPCR ligands: quests and promise
- PMID: 20136835
- PMCID: PMC2839259
- DOI: 10.1111/j.1476-5381.2009.00601.x
Rational design of dualsteric GPCR ligands: quests and promise
Abstract
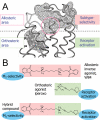
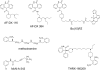
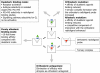
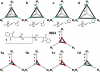
Dualsteric ligands represent a novel mode of targeting G protein-coupled receptors (GPCRs). These compounds attach simultaneously to both, the orthosteric transmitter binding site and an additional allosteric binding area of a receptor protein. This approach allows the exploitation of favourable characteristics of the orthosteric and the allosteric site by a single ligand molecule. The orthosteric interaction provides high affinity binding and activation of receptors. The allosteric interaction yields receptor subtype-selectivity and, in addition, may modulate both, efficacy and intracellular signalling pathway activation. Insight into the spatial arrangement of the orthosteric and the allosteric site is far advanced in the muscarinic acetylcholine receptor, and the design of dualsteric muscarinic agonists has now been accomplished. Using the muscarinic receptor as a paradigm, this review summarizes the way from suggestive evidence for an orthosteric/allosteric overlap binding to the rational design and experimental validation of dualsteric ligands. As allosteric interactions are increasingly described for GPCRs and as insight into the spatial geometry of ligand/GPCR-complexes is growing impressively, the rational design of dualsteric drugs is a promising new approach to achieve fine-tuned GPCR-modulation.
Figures

References
-
- Antony J, Kellershohn K, Mohr-Andrä M, Kebig A, Prilla S, Muth M, et al. Dualsteric GPCR targeting: a novel route to binding and signaling pathway selectivity. FASEB J. 2009;23:442–450. - PubMed
-
- Avlani VA, Gregory KJ, Morton CJ, Parker MW, Sexton PM, Christopoulos A. Critical role for the second extracellular loop in the binding of both orthosteric and allosteric G protein-coupled receptor ligands. J Biol Chem. 2007;282:25677–25686. - PubMed
-
- Ballesteros JA, Weinstein H. Integrated methods for the construction of three-dimensional models and computational probing of structure-function relations in G protein-coupled receptors. Methods Neurosci. 1995;25:366–428.
-
- Birdsall NJ, Lazareno S. Allosterism at muscarinic receptors: ligands and mechanisms. Mini Rev Med Chem. 2005;5:523–543. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials

