Hypoxia-induced proliferation of human pulmonary microvascular endothelial cells depends on epidermal growth factor receptor tyrosine kinase activation
- PMID: 20139181
- PMCID: PMC2853344
- DOI: 10.1152/ajplung.00122.2009
Hypoxia-induced proliferation of human pulmonary microvascular endothelial cells depends on epidermal growth factor receptor tyrosine kinase activation
Abstract
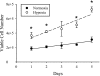
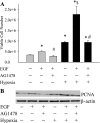
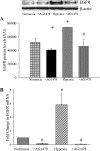
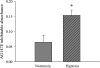
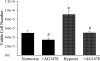
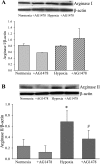
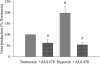
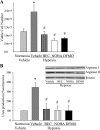
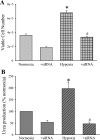
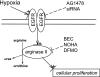
We hypothesized that hypoxia would activate epidermal growth factor receptor (EGFR) tyrosine kinase, leading to increased arginase expression and resulting in proliferation of human pulmonary microvascular endothelial cell (hPMVEC). To test this hypothesis, hPMVEC were incubated in normoxia (20% O(2), 5% CO(2)) or hypoxia (1% O(2), 5% CO(2)). Immunoblotting for EGFR and proliferating cell nuclear antigen was done, and protein levels of both total EGFR and proliferating cell nuclear antigen were greater in hypoxic hPMVEC than in normoxic hPMVEC. Furthermore, hypoxic hPMVEC had greater levels of EGFR activity than did normoxic hPMVEC. Hypoxic hPMVEC had a twofold greater level of proliferation compared with normoxic controls, and this increase in proliferation was prevented by the addition of AG-1478 (a pharmacological inhibitor of EGFR). Immunoblotting for arginase I and arginase II demonstrated a threefold induction in arginase II protein levels in hypoxia, with little change in arginase I protein levels. The hypoxic induction of arginase II protein was prevented by treatment with AG-1478. Proliferation assays were performed in the presence of arginase inhibitors, and hypoxia-induced proliferation was also prevented by arginase inhibition. Finally, treatment with an EGFR small interfering RNA prevented hypoxia-induced proliferation and urea production. These findings demonstrate that hypoxia activates EGFR tyrosine kinase, leading to arginase expression and thereby promoting proliferation in hPMVEC.
Figures
References
-
- Albina J, Mahoney E, Daley J, Reichner JS. Macrophage arginase regulation by CCAAT/enhancer-binding protein β. Shock 23: 168–172, 2005 - PubMed
-
- Bachetti T, Comini L, Francolini G, Bastianon D, Valetti B, Cadei M, Grigolato P, Ferrari R. Arginase pathway in human endothelial cells in pathophysiological conditions. J Mol Cell Cardiol 37: 515–523, 2004 - PubMed
-
- Banks M, Gerasimovskaya E, Tucker D, Stenmark K. Egr-1 antisense oligonucleotides inhibit hypoxia-induced proliferation of pulmonary artery adventitial fibroblasts. J Appl Physiol 98: 732–738, 2005 - PubMed
-
- Bivalacqua TJ, Burnett AL, Hellstrom WJ, Champion HC. Overexpression of arginase in the aged mouse penis impairs erectile function and decreases eNOS activity: influence of in vivo gene therapy of anti-arginase. Am J Physiol Heart Circ Physiol 292: H1340–H1351, 2007 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

