SLC22A2 gene 808 G/T variant is related to plasma lactate concentration in Chinese type 2 diabetics treated with metformin
- PMID: 20139901
- PMCID: PMC4002837
- DOI: 10.1038/aps.2009.189
SLC22A2 gene 808 G/T variant is related to plasma lactate concentration in Chinese type 2 diabetics treated with metformin
Abstract
Aim: To investigate the potential relationship between the SLC22A2 gene polymorphism and blood lactate concentration in Shanghai Hans suffering from type 2 diabetes mellitus (T2DM).
Methods: The SLC22A2 single nucleotide polymorphism (SNP) 808G/T was genotyped in 400 T2DM patients, including a metformin-treated group (n=200) and a non-metformin-treated group (n=200). Fasting plasma lactic acid levels were measured with an enzyme-electrode assay. Biochemical indexes, including plasma alanine aminotransferase (ALT), creatinine (Cr), and glycolated hemoglobin (HbA1c), were also measured.
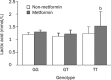
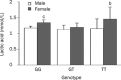
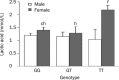
Results: The fasting plasma lactate concentration in the metformin-treated group was significantly higher than that in the non-metformin-treated group (1.29+/-0.45 mmol/L vs 1.18+/-0.44 mmol/L, P=0.015). Additionally, the ratio of patients with hyperlactacidemia was 8% (16/200) for the metformin-treated group and 5.5% (11/200) for the non-metformin-treated group, with no lactic acidosis found in either group. The frequency of the SLC22A2 808G/T T allele was 12.9%. Patients with the mutant genotype (TT) had a higher blood lactate concentration in the metformin-treated group than those in the non-metformin-treated group (t=2.492, P=0.013). This trend was not observed in the GG and GT genotypes when compared with metformin-treated and non-metformin-treated groups. Patients with the mutant genotype (TT) in the metformin-treated group also had a higher incidence of hyperlactacidemia compared with the GG genotype (40.0% vs 6.9%, P=0.050) in the metformin-treated group and the GG (6.0%, P=0.042) or GT (4.3%, P=0.043) genotypes in the non-metformin-treated group. In the metformin-treated group, there were significant gender differences in lactate concentrations in the TT (2.18+/-0.15 vs 1.04+/-0.27 mmol/L, P=0.008) and GG genotypes (1.40+/-0.51 vs 1.19+/-0.35 mmol/L, P=0.004). The lactate levels of women with the TT genotype were the highest in the metformin-treated group, but differences in lactate levels among the genotypes were not observed in the non-metformin-treated group.
Conclusion: There is an 808G/T polymorphism in the SLC22A2 gene in Chinese Hans with T2DM. The 808G>T variance in the SLC22A2 gene can affect the plasma lactate level and the incidence of hyperlactacidemia in T2DM patients undergoing metformin therapy. Additionally, the female patients carrying the TT genotype are prone to lactatemia.
Figures

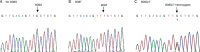
Similar articles
-
Polymorphism of organic cation transporter 2 improves glucose-lowering effect of metformin via influencing its pharmacokinetics in Chinese type 2 diabetic patients.Mol Diagn Ther. 2015 Feb;19(1):25-33. doi: 10.1007/s40291-014-0126-z. Mol Diagn Ther. 2015. PMID: 25573751 Clinical Trial.
-
Relationship of plasma creatinine and lactic acid in type 2 diabetic patients without renal dysfunction.Chin Med J (Engl). 2009 Nov 5;122(21):2547-53. Chin Med J (Engl). 2009. PMID: 19951568
-
The Effect of Genetic Variants of SLC22A2 (rs662301 and rs315978) on the response to Metformin in type 2 Saudi diabetic patients.Gene. 2024 Nov 15;927:148648. doi: 10.1016/j.gene.2024.148648. Epub 2024 Jun 7. Gene. 2024. PMID: 38852696
-
Association between organic cation transporter genetic polymorphisms and metformin response and intolerance in T2DM individuals: a systematic review and meta-analysis.Front Public Health. 2023 Jul 21;11:1183879. doi: 10.3389/fpubh.2023.1183879. eCollection 2023. Front Public Health. 2023. PMID: 37546319 Free PMC article.
-
Clinical pharmacokinetics of metformin.Clin Pharmacokinet. 2011 Feb;50(2):81-98. doi: 10.2165/11534750-000000000-00000. Clin Pharmacokinet. 2011. PMID: 21241070 Review.
Cited by
-
The role of pharmacogenetics in drug disposition and response of oral glucose-lowering drugs.Clin Pharmacokinet. 2013 Oct;52(10):833-54. doi: 10.1007/s40262-013-0076-3. Clin Pharmacokinet. 2013. PMID: 23719679 Review.
-
Mitochondrial related variants associated with cardiovascular traits.Front Physiol. 2024 Aug 27;15:1395371. doi: 10.3389/fphys.2024.1395371. eCollection 2024. Front Physiol. 2024. PMID: 39258111 Free PMC article.
-
Pharmacogenomics in Asian Subpopulations and Impacts on Commonly Prescribed Medications.Clin Transl Sci. 2020 Sep;13(5):861-870. doi: 10.1111/cts.12771. Epub 2020 Apr 13. Clin Transl Sci. 2020. PMID: 32100936 Free PMC article. Review.
-
Different effect of testosterone and oestrogen on urinary excretion of metformin via regulating OCTs and MATEs expression in the kidney of mice.J Cell Mol Med. 2016 Dec;20(12):2309-2317. doi: 10.1111/jcmm.12922. Epub 2016 Jul 29. J Cell Mol Med. 2016. PMID: 27469532 Free PMC article.
-
Pharmacogenomics in type 2 diabetes: oral antidiabetic drugs.Pharmacogenomics J. 2016 Oct;16(5):399-410. doi: 10.1038/tpj.2016.54. Epub 2016 Jul 19. Pharmacogenomics J. 2016. PMID: 27432533 Review.
References
-
- Hundal RS, Inzucchi SE. Metformin: new understandings, new uses. Drugs. 2003;63:1879–94. - PubMed
-
- Hermann LS, Scherstén B, Bitzén PO, Kjellström T, Lindgärde F, Melander A. Therapeutic comparison of metformin and sulfonylurea, alone and in various combinations, a double- blind controlled study. Diabetes Care. 1994;17:1100–9. - PubMed
-
- Dresser MJ, Xiao G, Leabman MK, Gray AT, Giacomini KM. Interactions of N-tetraalkylammonium compounds and biguanides with a human renal organic cation tranporter (hOCT2) Pharm Res. 2002;19:1244–7. - PubMed
-
- Kimura N, Masuda S, Tanihara Y, Ueo H, Okuda M, Katsura T, Inui K, et al. Metformin is a superior substrate for renal organic cation tranporter OCT2 rather than hepatic OCT1. Drug Metab Pharmacokinet. 2005;20:379–86. - PubMed
-
- Motohashi H, Sakurai Y, Saito H, Masuda S, Urakami Y, Goto M, et al. Gene expression levels and immunolocalization of organic ion transproters in the human kidney. J Am Soc Nephrol. 2002;13:866–74. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

