Active fragments from pro- and antiapoptotic BCL-2 proteins have distinct membrane behavior reflecting their functional divergence
- PMID: 20140092
- PMCID: PMC2816717
- DOI: 10.1371/journal.pone.0009066
Active fragments from pro- and antiapoptotic BCL-2 proteins have distinct membrane behavior reflecting their functional divergence
Abstract
Background: The BCL-2 family of proteins includes pro- and antiapoptotic members acting by controlling the permeabilization of mitochondria. Although the association of these proteins with the outer mitochondrial membrane is crucial for their function, little is known about the characteristics of this interaction.
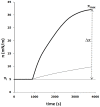
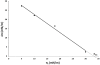
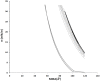
Methodology/principal findings: Here, we followed a reductionist approach to clarify to what extent membrane-active regions of homologous BCL-2 family proteins contribute to their functional divergence. Using isolated mitochondria as well as model lipid Langmuir monolayers coupled with Brewster Angle Microscopy, we explored systematically and comparatively the membrane activity and membrane-peptide interactions of fragments derived from the central helical hairpin of BAX, BCL-xL and BID. The results show a connection between the differing abilities of the assayed peptide fragments to contact, insert, destabilize and porate membranes and the activity of their cognate proteins in programmed cell death.
Conclusion/significance: BCL-2 family-derived pore-forming helices thus represent structurally analogous, but functionally dissimilar membrane domains.
Conflict of interest statement
Figures


Similar articles
-
Membrane-insertion fragments of Bcl-xL, Bax, and Bid.Biochemistry. 2004 Aug 31;43(34):10930-43. doi: 10.1021/bi036044c. Biochemistry. 2004. PMID: 15323553
-
Single-point mutations of a lysine residue change function of Bax and Bcl-xL expressed in Bax- and Bak-less mouse embryonic fibroblasts: novel insights into the molecular mechanisms of Bax-induced apoptosis.Cell Death Differ. 2011 Mar;18(3):427-38. doi: 10.1038/cdd.2010.112. Epub 2010 Oct 1. Cell Death Differ. 2011. PMID: 20885444 Free PMC article.
-
Humanin selectively prevents the activation of pro-apoptotic protein BID by sequestering it into fibers.J Biol Chem. 2020 Dec 25;295(52):18226-18238. doi: 10.1074/jbc.RA120.013023. Epub 2020 Oct 26. J Biol Chem. 2020. PMID: 33106313 Free PMC article.
-
Bid: a Bax-like BH3 protein.Oncogene. 2008 Dec;27 Suppl 1:S93-104. doi: 10.1038/onc.2009.47. Oncogene. 2008. PMID: 19641510 Review.
-
Structural biology of the Bcl-2 family of proteins.Biochim Biophys Acta. 2004 Mar 1;1644(2-3):83-94. doi: 10.1016/j.bbamcr.2003.08.012. Biochim Biophys Acta. 2004. PMID: 14996493 Review.
Cited by
-
µ-Calpain conversion of antiapoptotic Bfl-1 (BCL2A1) into a prodeath factor reveals two distinct alpha-helices inducing mitochondria-mediated apoptosis.PLoS One. 2012;7(6):e38620. doi: 10.1371/journal.pone.0038620. Epub 2012 Jun 20. PLoS One. 2012. PMID: 22745672 Free PMC article.
-
A lipocentric view of peptide-induced pores.Eur Biophys J. 2011 Apr;40(4):399-415. doi: 10.1007/s00249-011-0693-4. Epub 2011 Mar 26. Eur Biophys J. 2011. PMID: 21442255 Free PMC article. Review.
-
Apoptotic cell signaling in cancer progression and therapy.Integr Biol (Camb). 2011 Apr;3(4):279-96. doi: 10.1039/c0ib00144a. Epub 2011 Feb 22. Integr Biol (Camb). 2011. PMID: 21340093 Free PMC article. Review.
-
Bax-derived membrane-active peptides act as potent and direct inducers of apoptosis in cancer cells.J Cell Sci. 2011 Feb 15;124(Pt 4):556-64. doi: 10.1242/jcs.076745. Epub 2011 Jan 18. J Cell Sci. 2011. PMID: 21245196 Free PMC article.
-
Comparison of VILIP-1 and VILIP-3 binding to phospholipid monolayers.PLoS One. 2014 Apr 3;9(4):e93948. doi: 10.1371/journal.pone.0093948. eCollection 2014. PLoS One. 2014. PMID: 24699524 Free PMC article.
References
-
- Chipuk JE, Bouchier-Hayes L, Green DR. Mitochondrial outer membrane permeabilization during apoptosis: the innocent bystander scenario. Cell Death Differ. 2006;13:1396–1402. - PubMed
-
- Galonek HL, Hardwick JM. Upgrading the BCL-2 network. Nat Cell Biol. 2006;8:1317–1319. - PubMed
-
- Youle RJ, Strasser A. The BCL-2 protein family: opposing activities that mediate cell death. Nat Rev Mol Cell Biol. 2008;9:47–59. - PubMed
-
- Antonsson B, Montessuit S, Sanchez B, Martinou JC. Bax is present as a high molecular weight oligomer/complex in the mitochondrial membrane of apoptotic cells. J Biol Chem. 2001;276:11615–11623. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

