Staphylococcal PknB as the first prokaryotic representative of the proline-directed kinases
- PMID: 20140229
- PMCID: PMC2816222
- DOI: 10.1371/journal.pone.0009057
Staphylococcal PknB as the first prokaryotic representative of the proline-directed kinases
Abstract
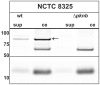
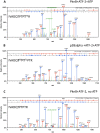
In eukaryotic cell types, virtually all cellular processes are under control of proline-directed kinases and especially MAP kinases. Serine/threonine kinases in general were originally considered as a eukaryote-specific enzyme family. However, recent studies have revealed that orthologues of eukaryotic serine/threonine kinases exist in bacteria. Moreover, various pathogenic species, such as Yersinia and Mycobacterium, require serine/threonine kinases for successful invasion of human host cells. The substrates targeted by bacterial serine/threonine kinases have remained largely unknown. Here we report that the serine/threonine kinase PknB from the important pathogen Staphylococcus aureus is released into the external milieu, which opens up the possibility that PknB does not only phosphorylate bacterial proteins but also proteins of the human host. To identify possible human targets of purified PknB, we studied in vitro phosphorylation of peptide microarrays and detected 68 possible human targets for phosphorylation. These results show that PknB is a proline-directed kinase with MAP kinase-like enzymatic activity. As the potential cellular targets for PknB are involved in apoptosis, immune responses, transport, and metabolism, PknB secretion may help the bacterium to evade intracellular killing and facilitate its growth. In apparent agreement with this notion, phosphorylation of the host-cell response coordinating transcription factor ATF-2 by PknB was confirmed by mass spectrometry. Taken together, our results identify PknB as the first prokaryotic representative of the proline-directed kinase/MAP kinase family of enzymes.
Conflict of interest statement
Figures

Similar articles
-
Transcriptome and functional analysis of the eukaryotic-type serine/threonine kinase PknB in Staphylococcus aureus.J Bacteriol. 2009 Jul;191(13):4056-69. doi: 10.1128/JB.00117-09. Epub 2009 Apr 17. J Bacteriol. 2009. PMID: 19376851 Free PMC article.
-
Expression and characterization of the Mycobacterium tuberculosis serine/threonine protein kinase PknB.Infect Immun. 1999 Nov;67(11):5676-82. doi: 10.1128/IAI.67.11.5676-5682.1999. Infect Immun. 1999. PMID: 10531215 Free PMC article.
-
The cell wall precursor lipid II acts as a molecular signal for the Ser/Thr kinase PknB of Staphylococcus aureus.Int J Med Microbiol. 2017 Jan;307(1):1-10. doi: 10.1016/j.ijmm.2016.12.001. Epub 2016 Dec 12. Int J Med Microbiol. 2017. PMID: 27989665
-
The impact of serine/threonine phosphorylation in Staphylococcus aureus.Int J Med Microbiol. 2010 Feb;300(2-3):137-41. doi: 10.1016/j.ijmm.2009.08.016. Epub 2009 Sep 23. Int J Med Microbiol. 2010. PMID: 19783479 Review.
-
Nuclear export of the stress-activated protein kinase p38 mediated by its substrate MAPKAP kinase-2.Curr Biol. 1998 Sep 24;8(19):1049-57. doi: 10.1016/s0960-9822(98)70442-7. Curr Biol. 1998. PMID: 9768359 Review.
Cited by
-
Proteomic discovery of host kinase signaling in bacterial infections.Proteomics Clin Appl. 2016 Oct;10(9-10):994-1010. doi: 10.1002/prca.201600035. Epub 2016 Sep 9. Proteomics Clin Appl. 2016. PMID: 27440122 Free PMC article. Review.
-
Deletion of a cation transporter promotes lysis in Streptococcus pneumoniae.Infect Immun. 2011 Jun;79(6):2314-23. doi: 10.1128/IAI.00677-10. Epub 2011 Mar 21. Infect Immun. 2011. PMID: 21422174 Free PMC article.
-
Metabolic conversion of CI-1040 turns a cellular MEK-inhibitor into an antibacterial compound.Sci Rep. 2018 Jun 14;8(1):9114. doi: 10.1038/s41598-018-27445-7. Sci Rep. 2018. PMID: 29904167 Free PMC article.
-
EssC is a specificity determinant for Staphylococcus aureus type VII secretion.Microbiology (Reading). 2018 May;164(5):816-820. doi: 10.1099/mic.0.000650. Epub 2018 Apr 5. Microbiology (Reading). 2018. PMID: 29620499 Free PMC article.
-
PrePhyloPro: phylogenetic profile-based prediction of whole proteome linkages.PeerJ. 2017 Aug 28;5:e3712. doi: 10.7717/peerj.3712. eCollection 2017. PeerJ. 2017. PMID: 28875072 Free PMC article.
References
-
- Rosqvist R, Forsberg A, Wolf-Watz H. Microinjection of the Yersinia YopE cytotoxin in mammalian cells induces actin microfilament disruption. Biochem Soc Trans. 1991;19:1131–1132. - PubMed
-
- Galyov EE, Hakansson S, Forsberg A, Wolf-Watz H. A secreted protein kinase of Yersinia pseudotuberculosis is an indispensable virulence determinant. Nature. 1993;361:730–732. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

