Warburg effect in chemosensitivity: targeting lactate dehydrogenase-A re-sensitizes taxol-resistant cancer cells to taxol
- PMID: 20144215
- PMCID: PMC2829492
- DOI: 10.1186/1476-4598-9-33
Warburg effect in chemosensitivity: targeting lactate dehydrogenase-A re-sensitizes taxol-resistant cancer cells to taxol
Abstract
Background: Taxol is one of the most effective chemotherapeutic agents for the treatment of patients with breast cancer. Despite impressive clinical responses initially, the majority of patients eventually develop resistance to Taxol. Lactate dehydrogenase-A (LDH-A) is one of the predominant isoforms of LDH expressed in breast tissue, which controls the conversion of pyruvate to lactate and plays an important role in glucose metabolism. In this study we investigated the role of LDH-A in mediating Taxol resistance in human breast cancer cells.
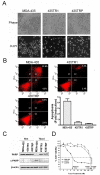
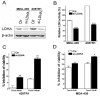
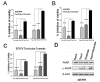
Results: Taxol-resistant subclones, derived from the cancer cell line MDA-MB-435, sustained continuous growth in high concentrations of Taxol while the Taxol-sensitive cells could not. The increased expression and activity of LDH-A were detected in Taxol-resistant cells when compared with their parental cells. The downregulation of LDH-A by siRNA significantly increased the sensitivity of Taxol-resistant cells to Taxol. A higher sensitivity to the specific LDH inhibitor, oxamate, was found in the Taxol-resistant cells. Furthermore, treating cells with the combination of Taxol and oxamate showed a synergistical inhibitory effect on Taxol-resistant breast cancer cells by promoting apoptosis in these cells.
Conclusion: LDH-A plays an important role in Taxol resistance and inhibition of LDH-A re-sensitizes Taxol-resistant cells to Taxol. This supports that Warburg effect is a property of Taxol resistant cancer cells and may play an important role in the development of Taxol resistance. To our knowledge, this is the first report showing that the increased expression of LDH-A plays an important role in Taxol resistance of human breast cancer cells. This study provides valuable information for the future development and use of targeted therapies, such as oxamate, for the treatment of patients with Taxol-resistant breast cancer.
Figures


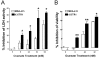
Similar articles
-
Targeting aspartate aminotransferase in breast cancer.Breast Cancer Res. 2008;10(5):R84. doi: 10.1186/bcr2154. Epub 2008 Oct 15. Breast Cancer Res. 2008. PMID: 18922152 Free PMC article.
-
Stable shRNA Silencing of Lactate Dehydrogenase A (LDHA) in Human MDA-MB-231 Breast Cancer Cells Fails to Alter Lactic Acid Production, Glycolytic Activity, ATP or Survival.Anticancer Res. 2017 Mar;37(3):1205-1212. doi: 10.21873/anticanres.11435. Anticancer Res. 2017. PMID: 28314283 Free PMC article.
-
Targeting lactate dehydrogenase‑A promotes docetaxel‑induced cytotoxicity predominantly in castration‑resistant prostate cancer cells.Oncol Rep. 2019 Jul;42(1):224-230. doi: 10.3892/or.2019.7171. Epub 2019 May 24. Oncol Rep. 2019. PMID: 31180564
-
Lactate dehydrogenase 5: an old friend and a new hope in the war on cancer.Cancer Lett. 2015 Mar 1;358(1):1-7. doi: 10.1016/j.canlet.2014.12.035. Epub 2014 Dec 17. Cancer Lett. 2015. PMID: 25528630 Review.
-
Lactate Dehydrogenase and its clinical significance in pancreatic and thoracic cancers.Semin Cancer Biol. 2022 Nov;86(Pt 2):93-100. doi: 10.1016/j.semcancer.2022.09.001. Epub 2022 Sep 9. Semin Cancer Biol. 2022. PMID: 36096316 Review.
Cited by
-
Warburg effect regulated by amphiregulin in the development of colorectal cancer.Cancer Med. 2015 Apr;4(4):575-87. doi: 10.1002/cam4.416. Epub 2015 Jan 30. Cancer Med. 2015. PMID: 25644309 Free PMC article.
-
Targeting Glucose Metabolism Enzymes in Cancer Treatment: Current and Emerging Strategies.Cancers (Basel). 2022 Sep 21;14(19):4568. doi: 10.3390/cancers14194568. Cancers (Basel). 2022. PMID: 36230492 Free PMC article. Review.
-
Mechanisms of Metabolic Reprogramming in Cancer Cells Supporting Enhanced Growth and Proliferation.Cells. 2021 Apr 29;10(5):1056. doi: 10.3390/cells10051056. Cells. 2021. PMID: 33946927 Free PMC article. Review.
-
Nutritional Regulation of Mammary Tumor Microenvironment.Front Cell Dev Biol. 2022 Feb 2;10:803280. doi: 10.3389/fcell.2022.803280. eCollection 2022. Front Cell Dev Biol. 2022. PMID: 35186923 Free PMC article. Review.
-
The Influence of Metabolism on Drug Response in Cancer.Front Oncol. 2018 Nov 2;8:500. doi: 10.3389/fonc.2018.00500. eCollection 2018. Front Oncol. 2018. PMID: 30456204 Free PMC article. Review.
References
-
- Tan M, Yu D. Molecular mechanisms of erbB2-mediated breast cancer chemoresistance. Adv Exp Med Biol. 2007;608:119–129. full_text. - PubMed
-
- Frankel Andrea, Buckman Robert, Kerbel Robert S. Abrogation of Taxol- induced G2-M Arrest and Apoptosis in Human Ovarian Cancer Cells Grown as Multicellular Tumor Spheroids. Cancer Res. 1997;57:2388–2293. - PubMed
-
- Chen LP, Cai SM, Fan JX, Li ZT. PEBA Regimen (Cisplatin, Etoposide, Bleomycin, and Adriamycin) in the Treatment of Drug-Resistant Choriocarcinoma. Gynecologic Oncology. 2002;56(2):231–234. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

