Single walled carbon nanotubes as reporters for the optical detection of glucose
- PMID: 20144355
- PMCID: PMC2771526
- DOI: 10.1177/193229680900300204
Single walled carbon nanotubes as reporters for the optical detection of glucose
Abstract
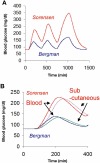
This article reviews current efforts to make glucose sensors based on the inherent optical properties of single walled carbon nanotubes. The advantages of single walled carbon nanotubes over traditional organic and nanoparticle fluorophores for in vivo-sensing applications are discussed. Two recent glucose sensors made by our group are described, with the first being an enzyme-based glucose sensor that couples a reaction mediator, which quenches nanotube fluorescence, on the surface of the nanotube with the reaction of the enzyme. The second sensor is based on competitive equilibrium binding between dextran-coated nanotubes and concanavalin A. The biocompatibility of a model sensor is examined using the chicken embryo chorioallantoic membrane as a tissue model. The advantages of measuring glucose concentration directly, like most optical sensors, versus measuring the flux in glucose concentration, like most electrochemical sensors, is discussed.
(c) 2009 Diabetes Technology Society.
Figures






References
-
- Diabetes atlas. 2nd ed. International Diabetes Federation; 2003.
-
- The Diabetes Control and Complications Trial Research Group. The effect of intensive treatment of diabetes on the development and progression of long-term complications in insulin-dependent diabetes-mellitus. N Engl J Med. 1993;329(14):977–986. - PubMed
-
- Barnett AH. Treating to goal: challenges of current management. Eur J Endocrinol. 2004;151(Suppl 2):T3–T7. discussion T29-30. - PubMed
-
- The absence of a glycemic threshold for the development of long-term complications. The perspective of the Diabetes Control and Complications Trial. Diabetes. 1996 Oct;45(10):1289–1298. - PubMed
-
- Wickramasinghe Y, Yang Y, Spencer SA. Current problems and potential techniques in in vivo glucose monitoring. J Fluoresc. 2004;14(5):513–520. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

