Neurological outcomes at 18 months of age after moderate hypothermia for perinatal hypoxic ischaemic encephalopathy: synthesis and meta-analysis of trial data
- PMID: 20144981
- PMCID: PMC2819259
- DOI: 10.1136/bmj.c363
Neurological outcomes at 18 months of age after moderate hypothermia for perinatal hypoxic ischaemic encephalopathy: synthesis and meta-analysis of trial data
Abstract
Objective: To determine whether moderate hypothermia after hypoxic-ischaemic encephalopathy in neonates improves survival and neurological outcome at 18 months of age.
Design: A meta-analysis was performed using a fixed effect model. Risk ratios, risk difference, and number needed to treat, plus 95% confidence intervals, were measured.
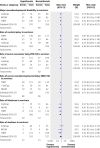
Data sources: Studies were identified from the Cochrane central register of controlled trials, the Oxford database of perinatal trials, PubMed, previous reviews, and abstracts. Review methods Reports that compared whole body cooling or selective head cooling with normal care in neonates with hypoxic-ischaemic encephalopathy and that included data on death or disability and on specific neurological outcomes of interest to patients and clinicians were selected. Results We found three trials, encompassing 767 infants, that included information on death and major neurodevelopmental disability after at least 18 months' follow-up. We also identified seven other trials with mortality information but no appropriate neurodevelopmental data. Therapeutic hypothermia significantly reduced the combined rate of death and severe disability in the three trials with 18 month outcomes (risk ratio 0.81, 95% confidence interval 0.71 to 0.93, P=0.002; risk difference -0.11, 95% CI -0.18 to -0.04), with a number needed to treat of nine (95% CI 5 to 25). Hypothermia increased survival with normal neurological function (risk ratio 1.53, 95% CI 1.22 to 1.93, P<0.001; risk difference 0.12, 95% CI 0.06 to 0.18), with a number needed to treat of eight (95% CI 5 to 17), and in survivors reduced the rates of severe disability (P=0.006), cerebral palsy (P=0.004), and mental and the psychomotor developmental index of less than 70 (P=0.01 and P=0.02, respectively). No significant interaction between severity of encephalopathy and treatment effect was detected. Mortality was significantly reduced when we assessed all 10 trials (1320 infants; relative risk 0.78, 95% CI 0.66 to 0.93, P=0.005; risk difference -0.07, 95% CI -0.12 to -0.02), with a number needed to treat of 14 (95% CI 8 to 47).
Conclusions: In infants with hypoxic-ischaemic encephalopathy, moderate hypothermia is associated with a consistent reduction in death and neurological impairment at 18 months.
Conflict of interest statement
Competing interests: All authors have completed the Unified Competing Interest form at
Figures




Comment in
-
Hypoxic ischaemic encephalopathy in infants.BMJ. 2010 Feb 9;340:c397. doi: 10.1136/bmj.c397. BMJ. 2010. PMID: 20144982 No abstract available.
References
-
- Gunn AJ, Gunn TR. The ‘pharmacology’ of neuronal rescue with cerebral hypothermia. Early Hum Dev 1998;53:19-35. - PubMed
-
- Eicher DJ, Wagner CL, Katikaneni LP, Hulsey TC, Bass WT, Kaufman DA, et al. Moderate hypothermia in neonatal encephalopathy: efficacy outcomes. Pediatr Neurol 2005;32:11-7. - PubMed
-
- Gluckman PD, Wyatt JS, Azzopardi D, Ballard R, Edwards AD, Ferriero DM, et al. Selective head cooling with mild systemic hypothermia after neonatal encephalopathy: multicentre randomised trial. Lancet 2005;365:663-70. - PubMed
-
- Shankaran S, Laptook AR, Ehrenkranz RA, Tyson JE, McDonald SA, Donovan EF, et al. Whole-body hypothermia for neonates with hypoxic-ischemic encephalopathy. N Engl J Med 2005;353:1574-84. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
