Human ELG1 regulates the level of ubiquitinated proliferating cell nuclear antigen (PCNA) through Its interactions with PCNA and USP1
- PMID: 20147293
- PMCID: PMC2856242
- DOI: 10.1074/jbc.M109.092544
Human ELG1 regulates the level of ubiquitinated proliferating cell nuclear antigen (PCNA) through Its interactions with PCNA and USP1
Abstract
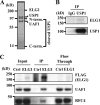
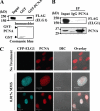
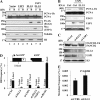
The level of monoubiquitinated proliferating cell nuclear antigen (PCNA) is closely linked with DNA damage bypass to protect cells from a high level of mutagenesis. However, it remains unclear how the level of monoubiquitinated PCNA is regulated. Here, we demonstrate that human ELG1 protein, which comprises an alternative replication factor C (RFC) complex and plays an important role in preserving genomic stability, as an interacting partner for the USP1 (ubiquitin-specific protease 1)-UAF1 (USP1-associated factor 1) complex, a deubiquitinating enzyme complex for PCNA and FANCD2. ELG1 protein interacts with PCNAs that are localized at stalled replication forks. ELG1 knockdown specifically resulted in an increase in the level of PCNA monoubiquitination without affecting the level of FANCD2 ubiquitination. It is a novel function of ELG1 distinct from its role as an alternative RFC complex because knockdowns of any other RFC subunits or other alternative RFCs did not affect PCNA monoubiquitination. Lastly, we identified a highly conserved N-terminal domain in ELG1 that was responsible for the USP1-UAF1 interaction as well as the activity to down-regulate PCNA monoubiquitination. Taken together, ELG1 specifically directs USP1-UAF1 complex for PCNA deubiquitination.
Figures


Similar articles
-
Is PCNA unloading the central function of the Elg1/ATAD5 replication factor C-like complex?Cell Cycle. 2013 Aug 15;12(16):2570-9. doi: 10.4161/cc.25626. Epub 2013 Jul 10. Cell Cycle. 2013. PMID: 23907118 Free PMC article. Review.
-
Deubiquitination of FANCD2 is required for DNA crosslink repair.Mol Cell. 2007 Dec 14;28(5):798-809. doi: 10.1016/j.molcel.2007.09.020. Mol Cell. 2007. PMID: 18082605 Free PMC article.
-
Regulation of the Fanconi anemia pathway by a SUMO-like delivery network.Genes Dev. 2011 Sep 1;25(17):1847-58. doi: 10.1101/gad.17020911. Genes Dev. 2011. PMID: 21896657 Free PMC article.
-
Dynamic regulation of PCNA ubiquitylation/deubiquitylation.FEBS Lett. 2011 Sep 16;585(18):2780-5. doi: 10.1016/j.febslet.2011.05.053. Epub 2011 Jun 1. FEBS Lett. 2011. PMID: 21640107 Free PMC article. Review.
-
The USP1/UAF1 complex promotes double-strand break repair through homologous recombination.Mol Cell Biol. 2011 Jun;31(12):2462-9. doi: 10.1128/MCB.05058-11. Epub 2011 Apr 11. Mol Cell Biol. 2011. PMID: 21482670 Free PMC article.
Cited by
-
SUMOylation of PCNA by PIAS1 and PIAS4 promotes template switch in the chicken and human B cell lines.Proc Natl Acad Sci U S A. 2018 Dec 11;115(50):12793-12798. doi: 10.1073/pnas.1716349115. Epub 2018 Nov 28. Proc Natl Acad Sci U S A. 2018. PMID: 30487218 Free PMC article.
-
Is PCNA unloading the central function of the Elg1/ATAD5 replication factor C-like complex?Cell Cycle. 2013 Aug 15;12(16):2570-9. doi: 10.4161/cc.25626. Epub 2013 Jul 10. Cell Cycle. 2013. PMID: 23907118 Free PMC article. Review.
-
A role for the yeast PCNA unloader Elg1 in eliciting the DNA damage checkpoint.Curr Genet. 2020 Feb;66(1):79-84. doi: 10.1007/s00294-019-01020-7. Epub 2019 Jul 22. Curr Genet. 2020. PMID: 31332476 Review.
-
The genome maintenance factor Mgs1 is targeted to sites of replication stress by ubiquitylated PCNA.Nucleic Acids Res. 2012 Jan;40(1):245-57. doi: 10.1093/nar/gkr738. Epub 2011 Sep 12. Nucleic Acids Res. 2012. PMID: 21911365 Free PMC article.
-
Anti-apoptotic effect by the suppression of IRF1 as a downstream of Wnt/β-catenin signaling in colorectal cancer cells.Oncogene. 2019 Aug;38(32):6051-6064. doi: 10.1038/s41388-019-0856-9. Epub 2019 Jul 10. Oncogene. 2019. PMID: 31292489
References
-
- Moldovan G. L., Pfander B., Jentsch S. (2007) Cell 129, 665–679 - PubMed
-
- Hoege C., Pfander B., Moldovan G. L., Pyrowolakis G., Jentsch S. (2002) Nature 419, 135–141 - PubMed
-
- Wang S. C., Nakajima Y., Yu Y. L., Xia W., Chen C. T., Yang C. C., McIntush E. W., Li L. Y., Hawke D. H., Kobayashi R., Hung M. C. (2006) Nat. Cell Biol. 8, 1359–1368 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

